- LOGIN
- MemberShip
- 2026-04-03 21:38:47
- Policy
- Bill to mandate generic substitutions gains momentum
- by Lee, Jeong-Hwan Sep 03, 2025 06:09am
- On September 2, the ruling party introduced a bill to mandate generic (ingredient-based) prescriptions for medicines with unstable supply. The bill establishes the legal basis for “shortage drugs” and allows prescribing by ingredient name instead of brand name. The bill holds significance in that it amends the Pharmaceutical Affairs Act to define drugs frequently in shortage and sets out legal procedures to designate such through public-private consultations, and amends the Medical Service Act to legislate the legitimacy and standards for mandatory ingredient prescriptions. In other words, the bill enforces a law that stipulates that “drugs in such unstable supply that generic prescribing must be enforced” through deliberations by a public-private consultative body. Representative Jong-tae Jang of the Democratic Party of Korea, who spearheaded the amendments, has stated he will push for their swift passage. The participation of fellow Democratic Party representative Yoon Kim (a physician) and Rebuilding Korea Party Rep Sun-min Kim (also a physician) is expected to reduce obstacles in upcoming committee reviews. Specifically, the bill requires the Ministry of Health and Welfare (MOHW) to establish a Supply Management Committee for Shortage Drugs. After deliberation and resolution by this committee, the Minister of Health and Welfare will designate shortage drugs. The committee will have up to 30 members, including the Vice Minister of Health and Welfare as Chair, Deputy Commissioner of the Ministry of Food and Drug Safety (MFDS) as Vice Chair. The remaining 28 will consist of : ▲Senior officials from relevant central government agencies (appointed by Presidential Decree), ▲ representatives recommended by the Chair of the Korean Pharmaceutical Association, ▲ representatives recommended by the Korean Medical Association under Article 28 of the Medical Service Act, ▲ representatives recommended by the Korea Pharmaceutical and Bio-Pharma Manufacturers Association and other relevant industry groups, as well as ▲ experts with sufficient knowledge and experience. Thus, government officials, pharmacists, physicians, manufacturers, and academia will collectively decide which drugs require mandatory generic prescribing due to unstable supply. The Minister of Health and Welfare will also have dedicated staff and budget authority to designate and de-designate shortage drugs, monitor supply conditions, and implement distribution improvement measures. The bill also legalizes distribution interventions alongside generic prescribing. If supply is deemed significantly disrupted, or upon request from another central administrative agency head, the Minister may—after committee review—order measures to improve distribution regarding sales outlets, procedures, volumes, and conditions. Pharmacies, medical institutions, wholesalers, and other designated entities will be legally obligated to comply with these measures. However, the Minister must first consult with the Minister of Strategy and Finance and the Chair of the Fair Trade Commission before issuing such orders. The MOHW Minister will also build and operate a shortage drug management system. This will allow requesting and collecting information necessary for distribution control, which includes production, shipments, sales, prescriptions, and dispensing data, from manufacturers, importers, wholesalers, pharmacies, and medical institutions. This effectively grants the MOHW authority over the entire supply chain, but also increases the Ministry’s accountability when shortages occur. The Minister may also designate certain shortage drugs as “emergency production/import drugs”, subject to committee review, which empowers the Minister to order manufacturers to produce or import the drugs. The Medical Service Act amendment stipulates that physicians and dentists must prescribe designated shortage drugs by generic name, not brand name. Violation will result in up to 5 years imprisonment or a fine up to KRW 50 million. This significant level of criminal penalty is expected to draw strong opposition from the Korean Medical Association and the broader medical community.
- Company
- Moderna’s latest variant-targeted COVID-19 vaccine approved
- by Whang, byung-woo Sep 03, 2025 06:08am
- Moderna Korea announced on the 1st that its LP.8.1 variant-targeted COVID-19 vaccine, ‘Spikevax LP Inj’, has been approved by the Ministry of Food and Drug Safety (MFDS). Spikevax LP has been confirmed to induce broad cross-immune responses against currently circulating variants, including the LP.8.1 strain, and it is authorized for use in adolescents aged 12 and above as well as adults. Moderna plans to supply the newly approved vaccine in time for the government’s 2025–2026 seasonal immunization program, which begins in October. The company noted that its COVID-19 vaccines have demonstrated strong immune effect and safety in large-scale Phase III clinical trials and extensive real-world evidence (RWE). A key feature is that elderly individuals aged 65 and above showed immune responses comparable to those in younger adults. Also, regardless of which vaccine type had been administered previously, the Moderna vaccine showed high immunogenicity when used as a subsequent dose. A domestic study conducted by the Korea Disease Control and Prevention Agency (KDCA) also confirmed that Moderna’s vaccine recorded the lowest breakthrough infection rate among the vaccines used in the early stages of the pandemic. According to the National Immunization Program guidelines, Spikevax LP will be provided free of charge to high-risk groups, including seniors aged 65 and over and residents of long-term care facilities. The Korean Society of Infectious Diseases has stressed that immunity acquired through infection or vaccination wanes over time, and with the emergence of new variants, periodic updated COVID-19 vaccination for high-risk groups is recommended. The LP.8.1 series vaccines supplied this season have already been recommended for use by the World Health Organization (WHO), the European Medicines Agency (EMA), and the U.S. Food and Drug Administration (FDA). Based on recommendations from its Vaccination Expert Committee, the KDCA decided to adopt the LP.8.1 vaccine, which demonstrated stronger neutralizing antibody responses compared to last season’s JN.1-based vaccines. Moderna is the only company manufacturing mRNA COVID-19 vaccines in Korea, through its partnership with Samsung Biologics, and continues to ensure a stable vaccine supply via ongoing cooperation with Boryung Biopharma. Sang Pyo Kim, General Manager of Moderna Korea, said, “COVID-19 remains a threat to high-risk groups, with hospitalizations on the rise for 7 consecutive weeks. Moderna is committed to delivering updated COVID-19 vaccines that address the latest variants in a timely manner, ensuring that people can be vaccinated safely.”
- Company
- 'Prevenar 20' now available at general hospitals
- by Eo, Yun-Ho Sep 02, 2025 06:11am
- Product photo of Prevenar 20 PFS The pneumococcal conjugate vaccine 'Prevenar 20,' which will soon be included in the National Immunization Program (NIP), is becoming available for prescription at general hospitals. According to industry sources, Pfizer Korea's Prevenar 20 has passed the drug committees (DC) of tertiary general hospitals, including Samsung Medical Center, Seoul National University Hospital, Asan Medical Center in Seoul, and Sinchon Severance Hospital, and medical institutes, including Kangnam Sacred Heart Hospital, Kyung Hee University Hospital at Gangdong, Pusan National University Hospital, Seoul National University Bundang Hospital, Ajou University Hospital, Pusan National University Yangsan Hospital, and Chungnam National University Hospital. Several of these hospitals, including Seoul National University Hospital, have approved codes for adult patients only. However, as Prevenar 20 will be included in the NIP effective October, more hospitals are expected to have pediatric vaccines. Prevenar 20 is a pneumococcal conjugate vaccine that was approved by the Ministry of Food and Drug Safety (MFDS) on October 31, 2024. Compared to the 13-valent vaccine, Prevenar 20 has added seven additional pneumococcal serotypes. Among domestically approved pneumococcal conjugate vaccines, it contains the most serotypes. In addition to existing serotype components in the 13-valent vaccine, Prevenar 20 contains seven additional serotypes (serotypes 1, 3, 4, 5, 6A, 6B, 7F, 8, 9V, 10A, 11A, 12F, 14, 15B, 18C, 19A, 19F, 22F, 23F, 33F). It can be used to prevent invasive diseases and pneumonia in all ages, including infants aged six weeks and above. Pneumococcus is a major bacterial pathogen that causes various diseases in infants and young children, including otitis media, pneumonia, and meningitis. Vaccination is crucial, especially as it can cause life-threatening invasive pneumococcal disease (IPD) in immunocompromised children. Despite South Korea's NIP supporting the 23-valent pneumococcal polysaccharide vaccine (PPSV23) for adults aged 65 and older, this age group has the highest incidence of invasive pneumococcal disease (IPD). From September 2014 to mid-November 2023, a total of 3,734 cases of IPD were reported. The incidence rate for adults aged 65 and over was 32.1 cases per 100,000 people, accounting for 54.8% of all cases. This rate is the highest among all age groups under 65 years old. The inclusion of Prevenar 20 in the NIP was decided after a comprehensive review by the Korea Expert Committee on Immunization Practices (KECIP) on the vaccine's safety, immunogenicity, and cost-effectiveness. Meanwhile, Korea Vaccine is responsible for the domestic promotion of pediatric Prevenar 20, while Chong Kun Dang handles the promotion of the adult vaccine.
- Policy
- Kanarb and Faslodex to keep their price until ruling
- by Lee, Tak-Sun Sep 02, 2025 06:11am
- The drug prices for Boryung's hypertension treatment ‘Kanarb’ and AstraZeneca's anticancer drug ‘Faslodex,’ which were determined to be reduced by the MOHW due to the entry of generics, will be maintained at their previous levels for now. The court has decided to suspend the execution of the price reduction until the first-instance ruling. As a result, the industry’s eyes are on whether these pharmaceutical companies will be able to halt the price reduction disposition through the main lawsuit. According to industry sources on the 1st, the Seoul Administrative Court accepted an application on the 27th of last month to suspend the execution of the price reduction order for 11 items, including Kanarb Tab. Consequently, the previous insurance price ceiling for the drugs will be maintained until two months after the date of the final judgment of the main trial. The affected items are Boryung's Kanarb Tab (3 dosages), Kanarb Plus Tab (2 dosages), Dukarb Tab (4 dosages), and Dongwha Pharmaceutical's LaCor Tab (2 dosages). At the end of June, the MOHW announced an ex officio adjustment and termination of the premium for these items following the entry of generic drugs containing the active ingredient, the single-ingredient fimasartan. Implementation was scheduled for July 1st. Four pharmaceutical companies' Kanarb generic products were listed for reimbursement last May. Consequently, health authorities proceeded with the ex officio price reduction process for Kanarb. Despite Boryung's objection, the price adjustment was ultimately finalized. The price caps for Kanarb Plus Tab and LaCor Tab, which are combination drugs that contain fimasartan, were also administratively reduced due to the price adjustment. For Dukarb, the premium granted for incrementally modified new drug combinations ended as two or more fimasartan-based single-ingredient drugs became available. Kanarb’s price was reduced by 30%, Dukarb by 21%, and Kanarb Plus and LaCor by 47%. Given that Kanarb and Dukarb each generate sales in the KRW 60 billion range, the price adjustment is expected to lead to a significant decrease in Boryung's overall sales performance. Consequently, many believed Boryung would seek to provisionally maintain its drug prices through litigation. The first trial for the main suit will commence in earnest with the first hearing scheduled for November 13. Kim & Chang is representing Boryung. The drug price for AstraZeneca's anticancer drug Faslodex (fulvestrant) will also be maintained until 30 days after the ruling date of the main case. With more than three companies producing generic versions, Faslodex's price adjustment period was scheduled to end last July. As a result, its insurance price cap was set to decrease from KRW 376,724 to KRW 288,194 starting August 1. AstraZeneca was reportedly considering withdrawing from the domestic market due to the Faslodex price cut disposition. This issue also surfaced during the confirmation hearing for Minister of MOHW Eun-Kyung Jeong. Domestic pharmaceutical companies have also expressed the view that if the original anticancer drug withdraws, it would be difficult for patients to find a substitute, given the market characteristics. Fortunately, the company did not withdraw its product but opted for litigation. On the 29th of last month, the Seoul Administrative Court granted its application for a stay of execution of the price reduction order. The main lawsuit was filed on July 25th, and no hearing date has been set yet. AstraZeneca's legal representative is the law firm Sejong. Although both pharmaceutical companies chose litigation to maintain drug prices, the risk is not entirely absent. This is because the Act on Drug Price Litigation Recovery and Refund that was implemented last year allows the National Health Insurance Service to refund the claimed amount for the trial period if the company loses the case. Nevertheless, analysis suggests the companies ultimately chose litigation due to the significant immediate financial loss and the potential for dispute over the price reduction decision.
- Company
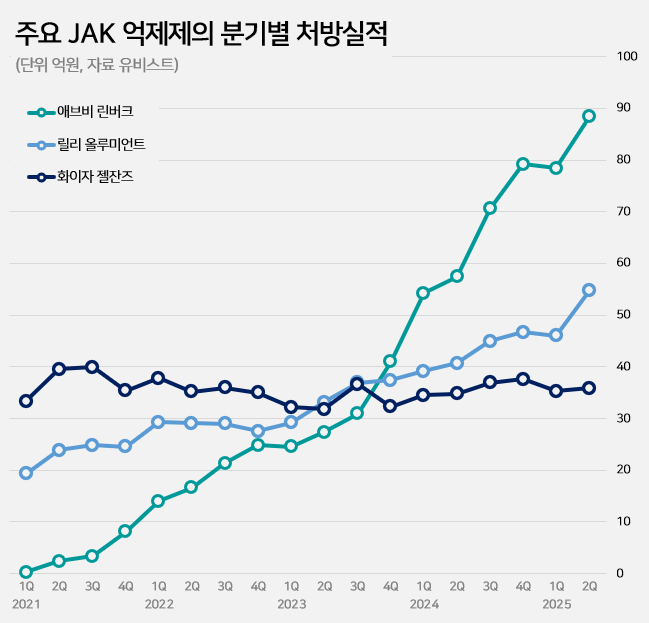
- Patent suits active to release Rinvoq generics
- by Kim, Jin-Gu Sep 02, 2025 06:10am
- Pic of Rinvoq The race to launch generics of AbbVie’s Rinvoq (upadacitinib), an oral treatment for autoimmune diseases, is showing signs of intensifying patent disputes. With the JAK inhibitor market expanding rapidly and Rinvoq solidifying its dominance, multiple companies are expected to mount patent challenges. According to industry sources on the 1st, Daewoong Pharmaceutical recently filed a passive scope confirmation trial against AbbVie, challenging the crystalline form patent of Rinvoq. Currently, two patents related to Rinvoq are registered: a substance patent expiring in May 2032 and a crystalline form patent expiring in October 2036. Daewoong's strategy is to first circumvent the crystalline form patent and then launch a generic version early, timed for the expiration of the substance patent. Chong Kun Dang opened the door to this dispute by challenging Rinvoq’s patents before Daewoong Pharmaceutical. Chong Kun Dang filed a related trial request on the 19th of last month. The pharmaceutical industry expects more companies to challenge Rinvoq’s patents from here on. One of the key requirements for obtaining first generic exclusivity is filing the initial invalidation trial. However, if another company files the same trial within 14 business days of the first filing date, it is also deemed to meet this requirement. This is why additional trial filings are expected in the coming weeks as companies move to secure first generic exclusivity for Rinvoq. According to UBIST, a pharmaceutical market research institution, outpatient prescriptions of JAK inhibitors in the first half of this year reached KRW 38.6 billion, up 40% from the KRW 27.5 billion in the same period last year. JAK inhibitors are experiencing rapid growth in the autoimmune disease treatment market, driven by the convenience of being oral formulations. The JAK inhibitor market, which was KRW 18.7 billion in 2020, grew by 36% to KRW 25.5 billion the following year. It continued to grow significantly each year: KRW 35.5 billion in 2022, KRW 40 billion in 2023, and KRW 62.2 billion in 2024. The expanded reimbursement granted for key products drove this market growth. Growth accelerated further last October when switching between JAK inhibitors for rheumatoid arthritis was allowed reimbursement. Major JAKi Prescriptions (AbbVie During this market expansion, Rinvoq has tightened its grip as the dominant player. In the first half of this year, Rinvoq recorded KRW 16.7 billion in prescriptions, a 49% increase from KRW 11.2 billion in the same period last year. Its growth has outpaced competitors such as Xeljanz (tofacitinib) and Olumiant (baricitinib). Having claimed the number one position in Q4 2023, Rinvoq has since expanded its market share, which now stands at 43% in the first half of this year. Rinvoq is a JAK inhibitor used for autoimmune diseases such as rheumatoid arthritis and atopic dermatitis. Its mechanism involves inhibiting inflammatory cytokines, thereby suppressing inflammation, pain, and cell activation.
- Company
- China's presence rises amid early-stage ADC candidate deals
- by Son, Hyung Min Sep 02, 2025 06:10am
- With competition in the antibody-drug conjugate (ADC) market intensifying, global deal trends are undergoing a clear shift. Transactions are now more active in the preclinical and early clinical stages, rather than in late-stage candidates, with collaborations between global pharma and Chinese companies standing out. Since the success of Enhertu, topoisomerase I (Topo I) payloads have effectively become the dominant approach. On the 29th, AimedBio and Samsung Medical Center held the 3rd ADC Conference at the hospital’s main auditorium, where AimedBio Chairman Nam-Gu Her highlighted recent changes in the ADC industry landscape. ADC therapies are novel anticancer drugs that link an antibody that is designed to bind a specific antigen on the surface of cancer cells with a cytotoxic payload through a linker. This design allows the drug to selectively target cancer cells, enhancing efficacy while minimizing adverse effects. According to Her, the ADC market has seen explosive growth since the launch of Enhertu in 2019. Over the past 5 years, deals worth USD 390 billion (KRW 540 trillion) have been completed, with more than 300 clinical trials initiated. The ADC Deal Boom and Trials Enhertu is a next-generation ADC composed of a monoclonal antibody that has the same structure as trastuzumab, which binds to receptors overexpressed on cancer cells and a highly potent Topo I inhibitor payload, linked via a tumor-selective cleavable linker. In a head-to-head study, Enhertu nearly doubled progression-free survival (PFS) compared to Kadcyla, which uses the same trastuzumab backbone but with a microtubule inhibitor payload. “Most companies are now developing ADCs with Topo I inhibitor payloads. In addition to Enhertu, Datroway, and Trodelvy are such examples,” said Her. In the past 5 years alone, 59 deals have been struck involving Topo I payloads, far outpacing tubulin inhibitors (28), degraders (11), immunomodulators (8), and DNA-damaging agents (5). Heo also noted that licensing dynamics have shifted. Historically, big deals centered on acquiring late-stage or commercial products. Today, however, activity has increased around preclinical candidates. High-profile examples made in the past include AstraZeneca’s partnerships on Enhertu and Datroway, AbbVie’s acquisition of Elahere, and Pfizer’s acquisition of Adcetris. However, nowadays, the same big pharma are actively scouting promising preclinical ADC assets. In the ADC payload development landscape, the use of topoisomerase I inhibitors has surged explosively. “The traditional deal trend was to acquire products rather than platforms. While deal sizes have decreased recently, interest in ADCs has not waned,” Her explained. “With many leading candidates already acquired or commercialized, transactions are now focusing on early-stage pipelines.” Chinese pharmaceutical companies are playing an increasingly prominent role. Technology exports from China surged from 55 cases in 2015 to over 300 in 2021, and recorded 213 last year. For instance, in 2022, MSD secured global rights to the TROP2 ADC sacituzumab tirumotecan from Kelun-Biotech for USD 1.41 billion. Last year, GSK obtained global rights to a B7-H3-targeting ADC from Hansoh Pharma. “The mainstream deal trend nowadays is acquiring on preclinical candidates through small-scale transactions. Global big pharma’s interest in Chinese firms is growing rapidly, reflecting China’s strengthened competitiveness in the ADC development scene.”
- Policy
- "MFDS' data-based drug shortage response"
- by Lee, Jeong-Hwan Sep 02, 2025 06:09am
- Jeong Eun Keong, Minister of Health and Welfare The Ministry of Health and Welfare (MOHW) announced that it will address drug shortages by utilizing information from the "Pharmaceutical Production, Supply, and Prescription Data-based Supply Risk Prediction System," which is currently being established by the Ministry of Food and Drug Safety (MFDS). The MOHW has also proposed drug shortage response strategies, including domestically producing the raw materials and sub-materials for medicines and vaccines, as well as expanding a production support program for drugs with unstable supply to include up to four pharmaceutical companies. The MOHW also stated that the introduction of abortion drugs could only be expedited after the scope of legal abortion is defined through amendments to the Mother and Child Health Act and the Criminal Act. The following Q&A is based on the MOHW's responses to written inquiries from People Power Party Representative Han Ji-a and Democratic Party of Korea Representative Nam In-soon of the National Assembly's Health and Welfare Committee on August 27. "Responding to unstable supply by linking production, supply, and prescription systems" The MOHW's Division of Pharmaceutical Policy and Division of Health Industry Promotion responded to Rep. Han Ji-ah's inquiries about policy plans to strengthen the drug supply chain. First, the MOHW stated that "securing a stable drug supply chain is essential for protecting and maintaining public life and health, and is an essential task directly related to health security." The Ministry explained that it is "monitoring drugs with supply interruptions or shortages in cooperation with the MFDS, and taking customized measures through a public-private consultative body when necessary." The MOHW also stated that it would create countermeasures by linking its system with the one being developed by the MFDS for drugs with unstable supply. The MFDS is currently building a data-based drug supply risk prediction support system to enhance the prediction of unstable supply situations. The system is designed to comprehensively analyze drug production, supply, and prescription information. In particular, the National Assembly has a pending amendment to the Pharmaceutical Affairs Act that would strengthen the linkage of information on drugs with unstable supply between the MOHW and the MFDS. The Ministry expressed its commitment to support this legislation to address drug shortages actively. The National Assembly also promised to make efforts to produce drug and vaccine raw materials and sub-materials domestically, support the production of drugs with unstable supply, and stockpile essential medicines. The plan is to expand the project that provides budget support for pharmaceutical companies producing drugs with unstable supply from one to four companies. To respond to human infections of avian influenza and bioterrorism, the Ministry plans to prioritize the introduction of the latest overseas vaccines and stockpile them with domestically developed vaccines after their development is complete. It will also focus on national cooperation to diversify the sources of imports for active pharmaceutical ingredients (APIs) from China and India. "Introducing abortion drug requires an amendment to both Mother and Child Health Act and the Criminal Act" The MOHW states that amendments to the Mother and Child Health Act and the Criminal Act are essential for the domestic approval of abortion drugs like Mifepristone. It stated that to create a safe abortion environment, the scope of abortion must be established through amendments to the Criminal Act. While the Constitutional Court's ruling in April 2019 that the abortion ban was unconstitutional effectively nullified the relevant penal provisions, a legislative void has persisted for over six years due to the failure to pass supplementary legislation. As a result, the MOHW's stance is that it is difficult to carry out abortion-related administrative tasks. The MOHW plans to work closely with relevant ministries, including the Ministry of Justice and the MFDS, to support future legal and institutional improvements. Minister Jeong Eun Keong gave the same response during a questioning session with People Power Party Representative Cho Bae-sook at the National Assembly's Special Committee on Budget and Accounts on August 27. Minister Jeong explained, "As the Criminal Act and Mother and Child Health Act have not been amended since the unconstitutional ruling, there are safety issues." She added, "While there are no approved drugs in Korea, most countries in the world use approved drugs." And added, "The World Health Organization (WHO) also provides recommendations based on gestational age." Minister Jeong said, "I will consult with the MFDS on safe usage methods. We will consider both the fetus's right to life and the woman's right to health in a balanced manner."
- Company
- How will fate fare for Roche’s two anticancer drugs?
- by Eo, Yun-Ho Sep 01, 2025 06:05am
- The industry’s eyes are on whether Roche's two anti-cancer drugs, which have faced repeated failures, will succeed in securing reimbursement listing this time. According to industry sources, Roche Korea's treatment for refractory diffuse large B-cell lymphoma(DLBCL), ‘Polivy (polatuzumab vedotin)’ and the PD-L1 inhibitor-based immunotherapy ‘Tecentriq (atezolizumab)’ passed the Health Insurance Review and Assessment Service's Cancer Disease Deliberation Committee last month and are currently under discussion for submission to the Drug Reimbursement Evaluation Committee. Both drugs failed to clear the Cancer Disease Deliberation Committee hurdle twice before finally passing on their third attempt. Therefore, it remains to be seen whether Roche, a pharmaceutical company specializing in anticancer drugs that has particularly struggled with Cancer Disease Deliberation Committee approvals since last year, can now achieve progress simultaneously in solid tumors and hematologic malignancies. Polivy originally aimed for reimbursement in 2021 for its first indication - third-line therapy in combination with BR therapy (bendamustine and rituximab), but failed to clear the CDDC hurdle. In the first half of 2023, Roche submitted a reimbursement application for Polivy’s use as first-line therapy in combination with R-CHP therapy (rituximab + cyclophosphamide, doxorubicin, prednisone). However, this application also faced rejection by the CDDC in February of last year. However, there is still hope. Polivy added the 60.9-month follow-up analysis results from the POLARIX study, which evaluated the efficacy of the Pola-R-CHP combination therapy as first-line treatment for DLBCL last year. In the case of Tecentriq, the drug’s reimbursement was first submitted to the Health Insurance Review and Assessment Service (HIRA) for review in May 2023, but it failed to set the reimbursement criteria. Its second attempt was again rejected by HIRA in July last year. At that time, Roche failed to secure reimbursement standards despite presenting additional results showing improved overall survival (OS) at the American Society of Clinical Oncology (ASCO) meeting. Meanwhile, the Polarix follow-up study of Polivy, presented at the American Society of Hematology (ASH) Annual Meeting 2024, is considered the first clinical trial in 20 years to expand the standard first-line treatment for DLBCL. Key results show that the Polivy combination therapy group demonstrated a clear improvement in overall survival (OS) compared to the control group treated with the existing standard therapy, R-CHOP. The lymphoma-related mortality rate was 9.0% in the Polivy combination therapy group and 11.4% in the R-CHOP control group. Approximately 5 years after treatment initiation, the risk of death in the Polivy combination therapy group decreased by 15%, an improvement over the previous 3-year follow-up result (6% risk reduction). In the case of Tecentriq, the 5-year follow-up study of IMpower010 showed that in patients with stage 2-3A non-small cell lung cancer (NSCLC) with PD-L1 expression ≥50%, who underwent complete resection and platinum-based chemotherapy, the Tecentriq adjuvant therapy arm resulted in an OS of 82.7%, which was significantly higher than that of the best supportive care (BSC) arm (65.3%).
- Opinion
- [Reporter's View] New opportunities have emerged for K-Bio
- by Son, Hyung Min Sep 01, 2025 06:04am
- In the global antibody-drug conjugate (ADC) market, the rapid rise of Chinese pharmaceutical companies is being noticed. Just five to six years ago, China was considered a 'technology follower.' However, with massive capital and swift clinical development, it has quickly become a key player in the global market. Chinese pharmaceutical companies are transforming the global landscape by establishing a comprehensive new drug development system, encompassing initial candidate discovery, clinical entry, late-stage trials, regulatory approval, and large-scale manufacturing. These companies are rapidly securing an increasing number of approvals in the US and Europe, while also inking major technology transfers with global pharmaceutical giants. The fact that Chinese companies can run their clinical and sales cycles on the strength of their domestic market alone is a point that Korean companies cannot easily match. With rapid clinical trials based on thousands of patient populations and strong policy support from regulatory authorities, China's 'volume offensive' is demonstrating immense power. In contrast, Korean ADC companies are exploring a different path. LigaChem Biosciences made major technology transfer deals with global pharmaceutical companies by developing a pipeline of over 20 candidates. Orum Therapeutics is accelerating its clinical development with a unique linker-payload technology. ABL Bio aims to expand into the ADC field based on its bispecific antibody platform, while IntoCell explores new ADC targets using its novel platform. In short, while China focuses on 'scale and speed,' Korea is emphasizing 'platform differentiation' with the global market in mind. This differentiated new drug development strategy is not just a competitive dynamic; it also opens up opportunities for collaboration. The production infrastructure and clinical networks of Chinese companies can serve as valuable resources for Korean firms. Indeed, some Korean companies are already trying to enter the market by partnering with Chinese Contract Research Organizations (CROs) to accelerate clinical trials or by forming strategic alliances with Chinese investors. Technology transfer also presents opportunities with Chinese pharmaceutical companies. For instance, LigaChem Biosciences transferred its technology to China's Fosun Pharma, and the HER2-targeted ADC from that deal is currently awaiting approval in China. However, collaboration with China doesn't come without risks. China's volume offensive can quickly drive down the price of a specific modality. With numerous antibodies, novel payloads, and diverse platforms entering the market simultaneously, competition is intensifying. From the perspective of global pharmaceutical companies, they will inevitably weigh the 'cost-effectiveness' of Korea versus China. If Korean companies' platform differentiation is not sufficiently proven, there is a significant risk that their technology will be viewed as a mere consumable commodity. Therefore, Korean companies' strategies must become more sophisticated. The companies need to secure differentiated data at each clinical stage, diversify partnerships with global pharmaceutical companies, and simultaneously manage a relationship of both 'competition' and 'collaboration' with China. Ultimately, how well they solidify their position in the US and European markets will determine the success or failure of the Korean ADC industry. China's rapid emergence in the pharmaceutical sector is both a threat and an opportunity for Korea. It has too much potential for collaboration to be dismissed as just a competitor, yet the risk of market encroachment is too significant to be viewed solely as a partner. The global challenge of the K-ADC will ultimately be judged by how it navigates this delicate balancing act. It is a critical time to clearly understand that even if Korea falls behind in a war of attrition and speed compared to China, there are still opportunities in platform differentiation and strategic partnerships.
- Company
- COVID-19 response more important for high-risk groups
- by Whang, byung-woo Sep 01, 2025 06:04am
- While COVID-19 has entered an endemic phase and become a resident disease in daily life, it remains a significant threat to high-risk groups like the elderly. In fact, the outbreak of COVID-19, which had slowed down for a while, has been showing an upward trend again. According to the Korea Disease Control and Prevention Agency's Infectious Disease Portal, signs of sporadic resurgence have emerged, such as the number of patients hospitalized for COVID-19 increasing from 139 to 220 within a week in early August. Hyun Jong Lee, Vice President of Academic Affairs, Korean Society of Otorhinolaryngology-Head and Neck Surgery (Director of Lee & Hong ENT Clinic), emphasized the importance of rapid diagnosis and treatment, stating, “Although public awareness has significantly declined since COVID-19 became endemic, the fatality rate remains high for high-risk patients if they become infected.” COVID-19 response remains critical for the elderly… proactive measures needed COVID-19 has shown a recurring pattern of outbreaks during the summer and winter seasons since entering the endemic phase. In clinical practice, the actual number of patients is reported to be higher than the official statistics. Lee explained, “Many hidden cases go undetected because COVID-19 is often mistaken for a common cold or air-conditioner-related illness. For those not considered high-risk, testing is not covered by insurance, so patients often don’t feel the need to get tested. In addition, many people who have had a previous infection assume, on their own, that ‘this time it can’t be COVID-19,’ which is not uncommon.” As a result, testing is carried out more actively only among patients aged 65 and older, those in poor general health, or when high-risk family members are living in the same household. Lee stated, “When recommending testing to suspected patients, only about 20-30% of those under 65 get tested, while about 50-70% of those aged 65 and older do.” He interpreted this as “likely because insurance covers diagnosis and treatment for those over 65, and they have greater health concerns.” Indeed, for those aged 60 and above, COVID-19 treatments are covered through reimbursement, allowing prescriptions to be issued upon a positive test result, which is why medical professionals actively recommend testing. However, for those under 60, both testing and treatment are out-of-pocket expenses unless specific risk factors exist, making it difficult for doctors to recommend them readily. Nevertheless, doctors unanimously agree that proactive measures are necessary for the elderly and those with underlying conditions who face a high risk of severe illness. According to Statistics Korea data, individuals aged 65 and older accounted for 91.9% of domestic COVID-19 deaths in 2022, and the fatality rate for those aged 65 and older was approximately 40 times higher than for those under 65. Lee emphasized, “For patients aged 65 and older, those with weakened immunity due to cancer, or those with serious underlying conditions, a COVID-19 infection still carries a very high risk of progressing to severe illness or death. When endoscopic examinations show clear signs that the condition may progress to bronchitis or pneumonia, physicians strongly recommend testing.” Paxlovid transforms COVID-19 response..."Contributing to Reduced Early Mortality" Currently, the mainstay of antiviral treatment for mild-to-moderate COVID-19 cases in Korea is Paxlovid (nirmatrelvir/ritonavir). Developed by Pfizer, this oral therapy has been proven effective when administered early in patients at high risk of severe disease progression. Lee evaluated, “Paxlovid is a very good drug that works by suppressing viral replication. It has demonstrated excellent effects when given in the early stages. Between 2023 and 2024, with stable supply and growing clinical experience, its use has expanded significantly, and we confirmed that it contributed to reducing early mortality.” The outstanding therapeutic effect of Paxlovid is also evident in patient experiences. According to Lee, “Elderly patients who had struggled greatly during previous COVID-19 infections now report a dramatic improvement after taking Paxlovid, saying they ‘feel much better.” Unlike when taking common cold medicine, patients reported feeling the effects directly after taking antiviral drugs, such as fever and pain subsiding much faster. These clinical observations are supported by research data. In the global EPIC-HR clinical trial, early administration of Paxlovid reduced the risk of hospitalization and death among COVID-19 patients by up to 86%. Similarly, a domestic study analyzing about 1.94 million patients reported that confirmed cases aged 60 or older who took Paxlovid within five days of symptom onset had a significantly lower risk of severe progression and death compared with those who did not. The prescription environment for oral COVID-19 treatments recently reached a major turning point. The government provided Paxlovid free of charge until last year, but starting this year, it transitioned to being reimbursed by national health insurance, meaning patients now bear part of the cost. Regarding this, Lee explained, “During the period when it was provided free of charge, some patients did not take the medication even after receiving a prescription because they didn't realize its value since they weren't paying for it. However, the shift from free to paid supply, introducing patient coinsurance, has brought one positive change: increased medication adherence.” “Reimbursement limited for the elderly and high-risk groups...Reimbursement criteria must be expanded” Nevertheless, this hasn't resolved all concerns on the site. Criticism persists that Paxlovid's current insurance reimbursement criteria are overly restrictive, potentially causing some high-risk patients to miss treatment opportunities. Lee emphasized, “Paxlovid currently faces many reimbursement restrictions. An infection that young family members might brush off lightly can be difficult for those in their 70s and 80s to recover from, potentially leading to death. Reimbursement should be lowered to include those aged 50 and above, and if safety is not an issue, it should also be expanded to include pediatric patients with underlying conditions.” In other words, experts believe that the current prescription system, centered on those aged 60 and above, should be expanded to include those in their 50s. They also advocate allowing healthcare providers to apply antiviral drugs at their discretion to middle-aged and older adults with underlying conditions. Finally, Lee urged high-risk COVID-19 patients and their families to adhere to two principles: prevention and prompt treatment. He advised, “As The Art of War states, ‘The best victory is one won without fighting,’ avoiding infection is paramount in COVID-19 management. Alongside hygiene protocols, establishing a primary defense line through vaccination in consideration of one's immune status is necessary.” Lee further added, “For those who cannot get vaccinated or fail to develop antibodies after vaccination, there is now Paxlovid. Since rapid diagnosis and treatment are crucial for its use, early testing and prompt treatment are a must for high-risk groups.”