- LOGIN
- MemberShip
- 2026-04-03 19:59:16
- Opinion
- [Reporter's View] Double standard against natural medicines
- by Kim, Jin-Gu Aug 22, 2025 06:06am
- The government announced the '4th New Natural Product Drug Development Promotion Plan' in May 2024. The plan outlines a joint effort by seven ministries, including the Ministry of Health and Welfare, the Ministry of Science and ICT, Ministry of Trade, Industry and Energy, Ministry of Environment, Ministry of Oceans and Fisheries, Rural Development Administration, and Korea Forest Service, to build a foundation for natural new drug R&D and promote its industrialization. To achieve this, the plan selected three strategies and six key tasks, and pledged support for customized consulting to enhance clinical success rates, as well as assistance for global expansion. The budget allocated for this was KRW 153 billion, a 2.7% increase from the previous year. However, in the reimbursement sector, the exact opposite policy is underway. A prime example is the ongoing discussion to delist the mugwort extract treatment for gastritis from reimbursement coverage. The government selected the mugwort extract as a target for the 2025 drug reimbursement appropriateness re-evaluation last year. In discussions held this year, the conclusion was that it 'lacks appropriateness for reimbursement.' While an objection procedure remains, the prevailing view, considering past precedents, is that a dramatic reversal of this decision is unlikely. On the one hand, the government is expanding the budget to industrially promote natural medicines, while on the other, it is reducing patient access due to a lack of clinical utility. This contradiction of simultaneously fostering industrial growth and removing a flagship product is seen as a decision that weakens the government's policy consistency. The problem of inconsistent policy is also evident in the differing results from two re-evaluations. The mugwort extract had already been recognized for its usefulness in a reimbursement re-evaluation conducted by health authorities 14 years ago. The Ministry of Health and Welfare reviewed the cost-effectiveness of five therapeutic categories, including cardiovascular and digestive ulcer drugs, as part of the 'Reorganization of the Listed Drugs List' in 2011. At that time, the usefulness of Stiren's 'gastritis treatment' indication was recognized. Conversely, its 'gastritis prevention' indication was questioned, and after a legal dispute over a delay in submitting clinical data, the conclusion was its removal from reimbursement. No issues were raised at the time regarding the usefulness of its gastritis treatment. This amounts to an opposite judgment being made on the same drug. This leaves questions about the consistency of policy not only in clinical settings but also from the perspective of pharmaceutical and biotech companies. A bigger problem is that this double standard has increased overall industry uncertainty and stifled research momentum. In the mid-2000s, major pharmaceutical and biotech companies maintained one or two natural medicine development pipelines; however, this is no longer happening. After Yungjin Pharmaceutical received approval for its atopic dermatitis treatment 'Yutoma' in 2012, no new natural medicines were produced in Korea for 13 years. Yutoma was never even launched due to cost issues, and its approval was canceled in 2022 for failing to submit re-evaluation data. In the same year, Chong Kun Dang received approval for its natural medicine 'G-Tec,' but it has not been launched due to delays in reimbursement listing. A field that was once highlighted as a national strategic project has been halted due to institutional contradictions. The controversy surrounding the mugwort extract extends beyond the survival of a few products and is directly linked to the trajectory of the Korean pharmaceutical and biotechnology industries. While past achievements cannot be uncritically praised, it would be a loss to dismiss the experience and assets accumulated in that process as an anachronistic product. Strengthening regulations to align with global standards is inevitable, but what is needed now is to correct this double standard. The contradiction of simultaneously promoting industrial growth and discussing delisting must be resolved. A balanced policy design that encompasses support for clinical research and commercialization, as well as quality standardization, should follow. Only then can natural medicines re-establish themselves as a competitive asset for the Korean pharmaceutical and bio-industry.
- Company
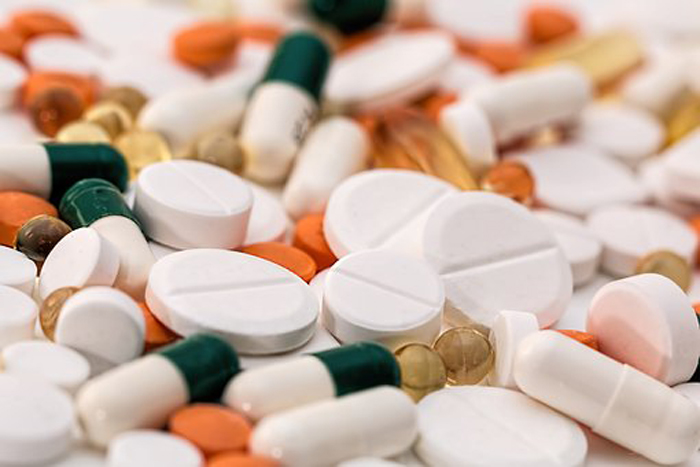
- Leclaza at global crossroads 1 year into FDA approval
- by Moon, sung-ho Aug 22, 2025 06:06am
- One year has passed since Leclaza (lazertinib) received FDA approval in combination with Johnson & Johnson’s Rybrevant (amivantamab). Expanding its influence beyond the United States to Europe and Asia, it has emerged as a global treatment option both in Korea and abroad. In the first half of this year, it recorded an overall survival rate (OS) exceeding 50 months, emerging as a global standard of care for non-small cell lung cancer and contributing to a major shift in the treatment paradigm. # With the FDA’s decision on whether to approve Rybrevant SC set to be determined in the second half of this year, another 'step-up' is also anticipated for Leclaza, depending on the result. According to industry sources on the 18th, the FDA approved the use of Rybrevant in combination with Leclaza (US brand name: Lazcluze) as a first-line treatment for adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) with EGFR exon 19 deletion or exon 21 L858R substitution mutation, or adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC). The combination was also approved in Europe in December of the same year, followed by the UK (March), Japan (March), and Canada (March) in the first quarter of this year, and China (July) in the second quarter. In May, prescriptions began in Japan, indicating the combination is gaining momentum in its expansion into the global market. Among the countries that have approved the combination, the combination’s performance in China is particularly noteworthy. With approximately 1 million new lung cancer diagnoses each year, lung cancer is the No.1 cancer in terms of incidence and mortality in China. This accounts for more than one-third of the 2.5 million new lung cancer cases worldwide. In addition, approximately 85% of lung cancer patients in China have non-small cell lung cancer (EGFR accounts for 40% of non-small cell lung cancer). Furthermore, the OS results of the Phase III (MARIPOSA) study presented at the European Lung Cancer Congress (ELCC 2025) held in Paris, France, in March this year have further accelerated the growth momentum. According to the Phase III MARIPOSA study, the combination therapy arm that received Rybrevant and Leclaza saw a 25% reduction in risk of death compared to the Tagrisso (osimertinib, AstraZeneca) arm (HR=0.75, 95% CI: 0.61–0.92, P
- Product
- Mounjaro arrives at pharmacies..₩310,000–350,000
- by Kang, Hye-Kyung Aug 21, 2025 06:07am
- Mounjaro (tirzepatide), which had been attracting considerable attention even before its arrival in Korea, has now been made available to pharmacies. With prescriptions also beginning on the 20th, pharmacies are bustling with activity. According to local pharmacies, two dosage forms, 2.5mg and 5mg, have begun arriving at pharmacies through wholesalers. Mounjaro Prefilled Pen 2.5mg, 5mg stocked at pharmacies A local pharmacist stated, “The 2.5mg and 5mg doses arrived this morning. Although no prescriptions have been made yet, we are receiving calls asking if the product has arrived. It seems that consumer interest is quite high.” The two main concerns for pharmacies are pricing and medication guidance. Mounjaro is priced in the ₩300,000 range, with the price lowest in Jongno at ₩290,000 ◆Prices vary between pharmacies, with the lowest price at KRW 290,000 = Since Mounjaro is a non-reimbursed drug, the price varies even between pharmacies. For this reason, pharmacies are struggling to set the initial price. According to telemedicine platforms, prices for 2.5mg in-hospital prescriptions at clinics range from KRW 320,000 to KRW 350,000 Pharmacies are pricing the medication between KRW 310,000 to KRW 350,000. While the number of pharmacies stocking Mounjaro is still limited, prices seem to be forming in the early to mid three hundred thousand won range. However, in Jongno, the price is set at KRW 290,000, which is the lowest price. ◆Medication guidance for Mounjaro?= Mounjaro comes in a package containing four single-use pre-filled pens. The outer case states, “Mounjaro is used once a week. Read the instructions carefully before using the medication and keep the instructions with the medication.” Mounjaro was approved as ▲an adjunct to diet and exercise therapy for improving blood glucose control in adult patients with type 2 diabetes; ▲an adjunct to low-calorie diet and exercise therapy for chronic weight management in adult patients; and ▲as an adjunct to a low-calorie diet and exercise therapy for the treatment of moderate-to-severe obstructive sleep apnea (OSA) in obese adult patients with an initial body mass index (BMI) of 30 kg/m2 or higher. When administered for weight management ▲Obese patients with an initial body mass index (BMI) of 30 kg/m2 or higher ▲Overweight patients with an initial body mass index (BMI) of 27 kg/m² or higher but less than 30 kg/m² and one or more weight-related comorbidities such as hypertension, dyslipidemia, type 2 diabetes, obstructive sleep apnea, or cardiovascular disease, may use the treatment. The recommended starting dose is 2.5 mg subcutaneous injection once weekly, increased to 5 mg subcutaneous injection once weekly after 4 weeks, and maintained at that dose. The maximum dose is 15 mg subcutaneous injection once weekly. If a dose is missed, administer the missed dose as soon as possible within 4 days (96 hours) of the missed dose. If more than 4 days have passed, skip the missed dose and administer the next dose on the scheduled day. Mounjaro can be administered at any time of the day, regardless of meals, and should be administered as a subcutaneous injection in the abdomen, thigh, or upper arm. It is recommended to rotate (alternate) the injection site with each dose. The recommended that the injections are refrigerated at 2–8°C. If necessary, each single-use pen can be stored at a temperature not exceeding 30°C for up to 21 days without refrigeration. Do not freeze. If frozen, the medication should not be used and should be stored in its original container to protect from light. The most commonly reported adverse reactions include gastrointestinal disorders such as nausea (very common), diarrhea (very common), and vomiting (common). Generally, the severity of these reactions was mild or moderate, and occurred more frequently during dose escalation, and decreased over time. Meanwhile, Lilly Korea announced that it plans to first release 2.5 mg and 5 mg doses in Korea, but will also supply higher doses of 7.5 mg, 10 mg, 12.5 mg, and 15 mg in accordance with patient demand. Meanwhile, the Ministry of Food and Drug Safety sent a notice to medical organizations and other relevant parties to prevent misuse of the drug following the release of Mounjaro. The MFDS emphasized, “Please prescribe and dispense the drug appropriately in accordance with the approved efficacy, dosage, and precautions for use. To prevent side effects and misuse, please provide accurate information about the drug (efficacy, dosage, and precautions for use) to the patients and instruct them on how to take the drug.”
- Company
- Nubeqa gains flexibility with indication expansion
- by Whang, byung-woo Aug 21, 2025 06:06am
- The influence of Nubeqa (darolutamide) is rising in the market with the company expanding its indication as a treatment for metastatic hormone-sensitive prostate cancer (mHSPC). Experts believe that the drug may settle as a flexible treatment option in Korea’s market as it has broadened its path as a personalized treatment. With the approval, overcoming the reimbursement hurdles is expected to serve as the key to competition in the future. Hyun-ho Han, professor of Urology at Severance HospitalBayer Korea held a media seminar on the 20th to celebrate the indication expansion for Nubeqa, an oral androgen receptor inhibitor (ARi), and highlighted its clinical significance. In June, Nubeqa was approved by the Ministry of Food and Drug Safety as part of a two-drug regimen in combination with androgen deprivation therapy (ADT) for the treatment of metastatic hormone-sensitive prostate cancer (mHSPC). In South Korea, Nubeqa was previously approved for use in combination with ADT and docetaxel for the treatment of mHSPC patients, as well as in combination with ADT for the treatment of high-risk non-metastatic castration-resistant prostate cancer (nmCRPC). With the expanded indication, Nubeqa can now be used not only as a three-drug regimen in combination with ADT and the chemotherapy agent docetaxel but also as a two-drug regimen in combination with ADT. The approval was based on the results of the global Phase III clinical trial ARANOTE, which evaluated the efficacy and safety of the two-drug regimen of Nubeqa in combination with ADT in 669 patients with mHSPC. The study results showed that the Nubeqa combination group significantly reduced the risk of radiographic progression or death by 46% compared to the placebo group, and this improvement in radiographic progression-free survival (rPFS) was consistently observed across all groups, including high-risk and low-risk mHSPC patients. Also, in the secondary endpoint, overall survival (OS), the Nubeqa combination therapy group demonstrated potential survival benefits compared to the placebo group, showing significant delays in disease progression in terms of PSA level, deterioration in quality of life, and pain progression, thereby proving clinically meaningful improvements in quality of life. The most significant aspect of this expanded indication is that Nubeqa is now the only treatment approved for both three-drug regimens with docetaxel and two-drug regimens with ADT in patients with metastatic hormone-sensitive prostate cancer. The introduction of this new option is expected to offer a more flexible treatment approach tailored to the individual condition and treatment goals of mHSPC, including providing treatment options for elderly patients or those who are not suitable for chemotherapy. Hyun-ho Han, professor of Urology at Severance Hospital, who presented at the media seminar, said, “Most mHSPC patients in Korea are elderly, aged 60 or older, and often have comorbidities. It is necessary to devise personalized treatment strategies based on clinical evidence, taking into account the patient's condition and preferences.” Professor Han added, “With the approval of the two-drug Nubeqa + ADT regimen, it is now possible to tailor treatment according to patient characteristics depending on whether docetaxel is used. Patients who require aggressive initial treatment can consider the three-drug combination therapy of Nubeqa + ADT + docetaxel, while patients with CNS concerns, such as the elderly or those with chronic diseases, or patients who are not suitable for docetaxel treatment, can consider the two-drug combination therapy of Nubeqa + ADT." Currently, Nubeqa is approved in over 85 countries worldwide as a treatment for mHSPC and nmCRPC, and as of 2024, it has surpassed annual sales of approximately KRW 2.4 trillion, establishing itself as a blockbuster drug. However, it is still non-reimbursed in Korea, resulting in high costs and limited access for patients. Currently, treatments such as Xtandi, Zytiga, and Erleada are reimbursed as treatments for mHSPC, so reimbursement is essential for competition with these products.. On this, Professor Han emphasized that Korea needs more treatment options when considering the criteria set for reimbursement of the existing drugs in prostate cancer. He said, “There are cases when patients with metastatic prostate cancer or non-metastatic castration-resistant prostate cancer are not eligible for reimbursement coverage. We want to offer cost-effective choices to patients, so any additional covered option that are introduced are welcome on our part.” Considering that existing treatments can only be used in combination with ADT, Professor Han believes that the three-drug regimen of Nubeqa + ADT + docetaxel may offer additional advantages. In addition, MyungKyu Noh, Oncology BU lead at Bayer Korea, said, “Bayer Korea is going through the reimbursement process so that domestic prostate cancer patients can promptly receive our treatment and its reimbursement as soon as possible. We will actively communicate with various stakeholders, including patients, medical staff, and the government, to improve access to treatment for all three indications.
- Company
- Reimb of polycythemia vera drug 'BESREMi' likely in Sept
- by Eo, Yun-Ho Aug 21, 2025 06:06am
- The polycythemia vera treatment 'BESREMi' is expected to be listed on the national health insurance list. The National Health Insurance Service (NHIS) and PharmaEssentia Korea have recently reached a final agreement on the drug price negotiation for BESREMi (ropeginterferon alfa-2b). As a result, if it passes the Health Insurance Policy Review Committee, it can be officially listed in September. BESREMi's company had previously proceeded with the reimbursement process for hydroxyurea-refractory or intolerant polycythemia vera in March 2023. It failed to pass the Health Insurance Review & Assessment Service (HIRA)'s Cancer Disease Review Committee in July of the same year. At that time, the Cancer Disease Review Committeedetermined that there was insufficient evidence to judge the clinical utility of BESREMi as a second-line treatment. PharmaEssentia subsequently supplemented the evidence for this drug's efficacy as a second-line therapy by adding domestic clinical data. It resubmitted the reimbursement application in March of last year, passed the Cancer Disease Review Committee in July of the same year, passed the Drug Reimbursement Evaluation Committee in May, and has now completed the negotiation stage. BESREMii is a next-generation interferon that selectively eliminates the JAK2 mutant gene, which is the cause of polycythemia vera. It was developed to improve the purity and tolerability of existing interferons, allowing for once-every-two-week administration for the initial 1.5 years and once every four weeks thereafter. BESREMi is currently recommended in the National Comprehensive Cancer Network (NCCN) and European LeukemiaNet (ELN) guidelines for the treatment of polycythemia vera, regardless of prior treatment history. Meanwhile, polycythemia vera is a rare blood cancer in which a somatic mutation in the bone marrow abnormally activates bone marrow function, leading to excessive production of red blood cells. According to HIRA data, the number of prevalent patients in Korea is around 5,000, and hydroxyurea is primarily used for more than half of these patients. However, the currently reimbursed drugs are not a fundamental treatment. For patients who fail hydroxyurea treatment, there is no new alternative, making it a disease with high unmet needs.
- Company
- Viatris signs exclusive sales and distribution deal for Brid
- by Whang, byung-woo Aug 21, 2025 06:05am
- Pic of Bridion Viatris Korea announced on the 20th that it has signed an exclusive domestic promotion and distribution agreement for the general anesthesia reversal agent Bridion (Sugammadex) through a strategic partnership with MSD Korea. Under the agreement, Viatris Korea officially took over the domestic promotion and distribution of Bridion as of the 7th of this month Bridion is a general anesthesia reversal agent developed by MSD, which was approved by the Ministry of Food and Drug Safety in October 2012 and launched in Korea in February 2013. Over the past decade, Bridion has established itself as a treatment option that enables rapid and predictable muscle relaxation recovery in clinical settings. Through this strategic partnership, Viatris Korea plans to expand its product portfolio into anesthesia in Korea, enhance patients' access to treatment, and strengthen its market leadership by leveraging its robust sales and marketing capabilities in the general hospital sector. Bill Schuster, Country Manager of Viatris Korea, said, “We are very pleased to add Bridion, MSD's general anesthesia reversal agent that has made many innovative marks in the anesthetic field, to our portfolio. This agreement will further solidify the partnership between the two companies and enable us to better provide treatment access to patients in need of Bridion amid rapidly changing market conditions.” He added, “As a company that empowers people to live healthier lives at every stage of life, Viatris Korea will continue to strive to provide healthcare professionals and patients with more effective and safer treatment options.” Bridion is a rapid and predictable neuromuscular blocking agent reversal drug that selectively reverses the effects of rocuronium or vecuronium, neuromuscular blocking agents used during anesthesia. It completely reverses neuromuscular blockade within an average of three minutes, enhancing the safety and efficiency of anesthesia recovery. Albert Kim, Managing Director of MSD Korea, said, “We expect that surgeons and patients in Korea will be able to continue to benefit from Bridion through our exclusive agreement with Viatris Korea, a company with strong expertise in the field of chronic diseases. Based on our trusted partnership, both companies will do their utmost to ensure a stable supply of Bridion in Korea.”
- Company
- Generics challenge the patent of mkt leading 'Rinvoq'
- by Kim, Jin-Gu Aug 21, 2025 06:05am
- Product photo of Rinvoq The patent challenges by generics targeting AbbVie's Janus kinase (JAK) inhibitor' Rinvoq (upadacitinib)' have begun. The pharmaceutical industry anticipates that patent challenges will further expand as Rinvoq strengthens its monopolistic position in the JAK inhibitor market, which is valued at approximately KRW 62 billion annually. Chong Kun Dang Files Invalidation Trial for Rinvoq's Methods of Manufacture…More Companies Expected to Join According to the pharmaceutical industry on the 20th, Chong Kun Dang filed a petition for trial to confirm the scope of rights against AbbVie on the 19th regarding Rinvoq's manufacturing method patent. This is the first patent challenge against Rinvoq. Rinvoq is a JAK inhibitor used to treat autoimmune diseases, including rheumatoid arthritis and atopic dermatitis. Its mechanism of action inhibits the production of inflammatory cytokines, thereby blocking inflammation, pain, and cell activation. After Xeljanz was launched in 2015, Olumiant and Rinvoq joined the competition in 2019 and 2021, respectively. Cibinqo and Jyseleca joined in 2023. Rinvoq has two registered patents: a substance patent expiring in May 2032 and a method of manufacture patent expiring in October 2036. It is expected that companies will first circumvent the patent expiring in 2036 and then launch their generics early, around the time of the expiration of the substance patent in 2032. The pharmaceutical industry expects that more companies will challenge Rinvoq's manufacturing method patent, following Chong Kun Dang. This is because the JAK inhibitor market is experiencing rapid growth, and Rinvoq is strenthening its monopolistic position. JAK Inhibitor According to pharmaceutical market research firm UBIST, the outpatient prescription sales of JAK inhibitors in the first half of this year were KRW 38.6 billion. This represents a 40% increase from the previous year, compared to KRW 27.5 billion in the first half of last year. JAK inhibitors are showing rapid growth in the autoimmune disease treatment market, driven by the convenience of their oral formulation. The JAK inhibitor market, which was KRW 18.7 billion in 2020, grew by 36% to KRW 25.5 billion the following year. The market size expanded with the addition of Rinvoq in 2021. Subsequently, it grew significantly each year, reaching KRW 35.5 billion in 2022, KRW 40 billion in 2023, and KRW 62.2 billion in 2024. During this period, the reimbursement expansion of key products drove market growth. An analysis suggests that the growth rate accelerated further after October 2024, when inter-class switching between JAK inhibitors for rheumatoid arthritis was approved for reimbursement. Rinvoq Strengthens Market Monopoly…H1 Market Share at 43% While the market has expanded, Rinvoq has further strengthened its monopolistic position. In the first half of this year, Rinvoq's prescription sales reached KRW 16.7 billion, a 49% increase from KRW 11.2 billion in the same period last year. Although prescription sales for other major JAK inhibitors also increased, their growth was not as rapid as Rinvoq's. Olumiant, the second-leading product in the market, saw a 26% increase from KRW 8 billion in the first half of last year to KRW 10.1 billion in the first half of this year. Xeljanz grew by 3% from KRW 6.9 billion to KRW 7.1 billion. Cibinqo and Jyseleca recorded prescription sales of KRW 3.2 billion and KRW 1.6 billion, respectively, in the first half of this year. As of the first half of this year, Rinvoq's market share in the JAK inhibitor market reached 43%. Compared to its 19% market share in the first half of 2022, this represents a 24 percentage point increase in three years. At the same time, Rinvoq, which was the third-largest player after Olumiant and Xeljanz, rose to the top spot in the market from the first half of 2024. Quarterly Sales Performance of Major JAK Inhibitors (unit: KRW 100 million, source: UBIST). GREEN-Rinvoq, SKY BLUE-Olumiant, BLUE-Xeljanz The rapid increase in Rinvoq's prescription sales is attributed to its having the most indications in Korea. Rinvoq holds indications for: ▲rheumatoid arthritis ▲psoriatic arthritis ▲ankylosing spondylitis ▲atopic dermatitis (adults and adolescents aged 12 and older) ▲ulcerative colitis and ▲Crohn's disease. Competing drug Olumiant has indications for: ▲rheumatoid arthritis ▲atopic dermatitis (adults and children aged 2 and older) ▲alopecia areata and ▲polyarticular juvenile idiopathic arthritis (pJIA). Xeljanz has indications for: ▲rheumatoid arthritis ▲psoriatic arthritis and ▲ankylosing spondylitis. Cibinqo has secured an indication for atopic dermatitis in adults and adolescents aged 12 and older, while Jyseleca has indications for rheumatoid arthritis and ulcerative colitis. A variable in this market is the launch of Xeljanz generics. The Xeljanz substance patent is set to expire in November this year. The crystal form patent, which expires in 2027, has been overcome by generic companies through numerous invalidation and circumvention trials. This means that Xeljanz generics are expected to launch all at once after November this year. Currently, 58 companies have obtained product approvals for 67 generic items and are ready for launch. Xeljanz generics are expected to be launched simultaneously without priority sales rights. This is because generic companies' victories in invalidation trials led to the deletion of the listed patents, making it impossible to acquire priority sales rights.
- Opinion
- [Desk View] Can NHI alone cover the costs of cancer drugs?
- by Lee, Tak-Sun Aug 20, 2025 06:23am
- The reimbursement claim amount submitted for anti-cancer drugs has been increasing robustly every year. According to the '2024 Pharmaceutical Reimbursement Claims Data' published by the Health Insurance Review & Assessment Service, drug expenditure for cancer diseases in 2024 amounted to KRW 4.1372 trillion, a 7.4% increase from KRW 3.8506 trillion in 2023. The rate of increase in cancer drug expenditures is steeper than the 4.5% increase for overall drug expenditures (KRW 26.9897 trillion). These data indicate that the costs of anti-cancer drugs are rising with the emergence of expensive drugs like immunotherapies and targeted therapies. The problem lies in what comes next. If the anti-cancer drugs currently awaiting reimbursement approval are approved, an additional KRW 2-3 trillion is expected to be added soon. The national health insurance, which is accumulating financial deficits, may no longer be able to cover these rising costs of anti-cancer drugs. Global pharmaceutical companies are increasingly combining new anti-cancer drugs or expanding investments in more expensive first-line treatments. As a result, cancer treatment is evolving. However, the latest high-cost anti-cancer drugs are waiting in a long queue for reimbursement, putting a burden on national health insurance finances. Some are suggesting that the 5% patient co-payment rate for anti-cancer drugs should be slightly increased. However, adjusting a patient's co-payment rate once it has been lowered is not an easy task. The government and politicians probably refrain from such a move, as it could cost them votes. For this reason, there is a growing argument that South Korea should also establish a separate fund for anti-cancer or rare disease drugs, similar to the UK, to save national health insurance finances and improve access to new drugs. However, the government does not seem to be acknowledging the seriousness of this issue yet. During the candidacy confirmation hearing, Jeong Eun Kyeong, Minister of Health and Welfare, expressed the view that expanding reimbursement coverage should be prioritized over creating a separate fund for patients with rare and severe diseases. The issue of national health insurance drug expenditures is not new, but the current instability differs from past patterns. In particular, merely adjusting the prices of drugs with expired patents, as in the past, has limited effectiveness in alleviating the financial pressure caused by the entry of high-cost drugs, such as innovative new anti-cancer drugs. It is also difficult for the government not to permit the entry of new, more effective anti-cancer drugs. The financial losses caused by these innovative anti-cancer drugs are, therefore, unavoidable. In this situation, it isn't easy to solve both financial stability and access to new drugs with past methods of drug cost reduction or reimbursement expansion. We hope that the new government realizes the seriousness of the cancer drug expenditure problem and prioritizes its policies.
- Opinion
- [Reporter's View] Innovation exists in treating all diseases
- by Eo, Yun-Ho Aug 20, 2025 06:23am
- When discussing Korea’s insurance reimbursement system, the qualifier “life-threatening” has long been a source of frustration. It is no exaggeration to say that, for the pharmaceutical industry, this phrase has been the number one target for removal since the very beginning. This was the case in determining eligibility for the Risk Sharing Agreement (RSA) and the exemption from pharmacoeconomic evaluation, and recently, even the preferential treatment for innovative drugs that allow the application of a flexible ICER threshold—another long-standing aspiration—has seen the phrase “life-threatening” implicitly applied in a different form. However, it is now necessary to question the very notion that a disease must be life-threatening to be considered serious. In particular, new drugs for chronic diseases, which are indirect causes of death for a large number of people due to comorbidities and complications, have long been left neglected in Korea’s reimbursement system. Although there are already many old drugs on the market and the number of new drugs being developed has decreased, the new drugs that represent a paradigm shift are being neglected. The breast cancer treatment Trodelvy was the first innovative drug to receive preferential treatment and ICER benefits, and was added to the reimbursement list in June. According to reports, the threshold for Trodelvy was set at KRW 70 million. This is an unprecedented amount. The Health Insurance Review and Assessment Service has stated that it does not use explicit thresholds, but it is well established that since the pharmacoeconomic evaluation system was first introduced in 2007 that the ICER threshold has been set at KRW 25 million for general drugs and KRW 50 million for anticancer drugs, based on the per capita GDP of KRW 25 million at the time, and has remained since for 18 years. In fact, according to HIRA's announcement last year, the ICER values for drugs submitted for pharmacoeconomic evaluation from 2019 to 2023 ranged from KRW 12.06 million to KRW 36.1 million for general drugs and KRW 25.88 million to KRW 47.92 million for anticancer drugs. This is why Trodelvy’s reimbursement listing is a significant milestone. However, such milestones should not become extremely rare cases. Providing benefits that cannot be applied in real life is meaningless. The criteria for receiving ICER benefits include three requirements, one of which states, “Significant clinical improvement must be recognized in final outcome measures such as extended survival.” Although the term “survival” implies “life-threatening,” it was not explicit. And in November last year, Kook-Hee Kim, Director of the Pharmaceutical Management Division at HIRA, said, “If innovativeness of a drug needs to be recognized in consideration of the severity of the disease and the social burden of the disease, the ICER threshold can be raised even if all three requirements are not met,” suggesting the possibility of flexible screening. In July, Minister Eun-kyung Jeong, who was appointed as the new Minister of Health and Welfare under President Jae-myung Lee’s administration, also mentioned the case of Trodelvy during her confirmation hearing in the National Assembly and expressed her agreement with the need for policy changes to recognize innovation. Under these circumstances, Mounjaro, which has been attracting public attention as a treatment for obesity, recently submitted a reimbursement application for its diabetes indication. While its weight-loss effect is well-known, Mounjaro's achievements in the diabetes field are also significant. Beyond blood sugar control targets, 6 out of 10 patients achieved normal blood sugar levels without an increased risk of hypoglycemia, reaching the ultimate treatment goals of preventing cardiovascular complications and reducing mortality. It even demonstrates the potential for “remission” in diabetes. However, assuming that the normal listing process is followed, it seems extremely unlikely that Mounjaro will be granted reimbursement for diabetes. As mentioned earlier, chronic diseases are already mainly treated with off-patent drugs. Given that these drugs are the comparators, it is difficult to predict a bright future for the entry of a new biopharmaceutical in the area. New drugs and drug prices are currently facing a critical turning point. Amidst the Trump administration's pressure on South Korea's drug pricing policy and the flood of high-priced drugs, concerns about “Korea passing” are growing, and future policy directions could have a significant impact on public health. With Korea already a foot into a super-aged society, it is crucial to make policy decisions from a long-term perspective to build a sustainable society capable of overcoming future health crises. We place our hopes in the Lee administration's pledge to “create a country where no one worries about illness.”
- Company
- Expanded patent dispute over cancer drug 'Xtandi'
- by Kim, Jin-Gu Aug 20, 2025 06:22am
- Product photo of Xtandi The number of companies challenging the patent for Astellas' prostate cancer treatment, 'Xtandi (enzalutamide)', has expanded to six. Attention has been drawn to the fact that major pharmaceutical companies, such as Chong Kun Dang, Hanmi Pharmaceutical, and JW Pharmaceutical, have joined this latest patent challenge. According to the pharmaceutical industry on August 19, Hanmi Pharmaceutical filed a petition for trial to confirm the scope of rights against Astellas for Xtandi's composition patent on August 18. As a result, the total number of companies that have filed circumvention trials against Xtandi's composition patent has increased to six. Earlier, Alvogen Korea was the first to file the trial on the 1st of this month, followed by Chong Kun Dang, JW Pharmaceutical, GL Pharma, and Kuhnil, where all filed the same trial. The pharmaceutical industry is paying attention to the fact that major pharmaceutical companies, such as Chong Kun Dang, Hanmi Pharmaceutical, and JW Pharmaceutical, have successively joined this patent challenge. This is because there have not been many cases where major pharmaceutical companies have simultaneously challenged patents for anti-cancer drugs. Typically, there is a high preference for prescribing original anti-cancer drugs, especially in large hospitals. Domestic pharmaceutical companies, which have strong sales capabilities for chronic disease treatments in clinics, have been hesitant to challenge patents for anti-cancer drugs for this reason. Furthermore, even if they succeeded in a patent challenge and launched a generic, the profits generated were not significant, making major pharmaceutical companies even more reluctant. There have been cases of patent challenges for liver cancer treatments like 'Lenvima (lenvatinib),' 'Cabometyx (cabozantinib),' and breast cancer treatment 'Ibrance (palbociclib).' However, these were sporadic challenges primarily led by Boryung, which has a separate oncology business unit, or Samyang Biopharm, which specializes in the generic oncology business. In the case of the oncology drug patent, Hanmi Pharmaceutical was the only company challenging the 'Nexavar (sorafenib) patent.' Meanwhile, in the patent challenge for the prostate cancer treatment Xtandi, major pharmaceutical companies like Chong Kun Dang, Hanmi Pharmaceutical, and JW Pharmaceutical have successively filed trials. If they win this trial, generic competition is expected to intensify after the substance patent expires in June 2026. Xtandi is Astellas' prostate cancer treatment, protected by two patents: a substance patent that expires in June 2026 and a composition patent that expires in September 2033. The plan for the generic companies is to circumvent the composition patent, which expires in 2033, and then launch their generics early around the expiration of the substance patent in 2026. Astellas received approval for Xtandi Soft Cap in 2013. Last year, it added two dosages of Xtandi tablets. Xtandi competes with Janssen's 'Erleada (apalutamide)' and 'Zytiga (abiraterone)' in the first-line prostate cancer treatment market. Recently, 'Akeega (abiraterone·niraparib),' a new first-line treatment, was launched. Akeega is a drug that combines Zytiga with Takeda's Zejula (niraparib). Recent trends indicate that while Zytiga's sales are declining following the launch of generics, Xtandi and Erleada are on an upward trend. According to pharmaceutical market research firm IQVIA, Xtandi's sales increased by 1.9 times in four years, from KRW 23 billion in 2019 to KRW 43.2 billion in 2023. It is estimated to have generated sales of around KRW 50 billion last year.