- LOGIN
- MemberShip
- 2026-04-03 19:58:45
- Policy
- Periodic drug price system reform included as national task
- by Lee, Jeong-Hwan Aug 18, 2025 06:02am
- President Lee Jae-myung attended the National Planning Committee The government has selected a policy to improve the drug price calculation system and establish a periodic drug price adjustment mechanism as part of its national policy task, with the goal of ensuring the sustainability of health insurance, and is pushing ahead with its implementation. Depending on the specific details of the national policy task to be established in the future, it is noteworthy that this could lead to additional post-marketing management regulations, such as a price reduction mechanism for generic drugs with expired patents, which is a highly concentrated area in the domestic pharmaceutical industry. To build a primary care-based health and care system, the government plans to gradually expand the local primary care physician model starting next year (2026) and adopt the institutionalization of telemedicine and public electronic prescription transmission systems as national policy tasks through social consensus. According to the National Assembly and the medical community on the 13th, the National Planning Committee has adopted such policy directions as national policy tasks. Transition to a sustainable healthcare system The National Planning Committee proposed improving the drug price calculation system and establishing a periodic drug price adjustment mechanism as a means of stabilizing the sustainability of the national health insurance budget, in order to curb expenditure factors that worsen the healthcare system, such as budget leakage. The government has been managing national health insurance drug expenses by adjusting the insurance ceiling price (drug price) of new and generic drugs through the post-marketing drug management system. In particular, following research conducted last year by Professor Dong-sook Kim’s research team at Kongju National University under the Yoon administration, the Ministry of Health and Welfare recently commissioned and launched additional research on the generic drug price system following the election of President Jae-myung Lee and the appointment of MOHW Minister Eun-kyung Jeong. The ministry has entered into a direct contract with a research team led by Professor Seon-mi Jang of the College of Pharmacy at Gachon University to conduct a study titled “Reform of the Drug Price Model (Focusing on Generic Drugs).” The results of this study is expected to serve as a basis for the government's drug price reductions. The Ministry of Health and Welfare under the Lee administration also agrees that it is necessary to establish financial leeway for national health insurance by adjusting the prices of old drugs with uncertain efficacy, so there is great social interest in whether new drug price reduction mechanisms, such as the overseas reference pricing reevaluation measures will emerge. However, there are also opinions that it is difficult to judge whether generic drug prices in South Korea are high or low compared to other developed countries. The Korean pharmaceutical industry is dissatisfied that the government is focused solely on establishing policies to reduce drug prices and is relatively neglecting policies that award premiums on drug prices. Ultimately, the pharmaceutical industry is expected to respond depending on how the details and direction of the national policy agenda are decided. Expansion of telemedicine and establishment of a public electronic prescription system The National Planning Committee has also selected the promotion of public health through primary care-based health management as a national policy task. The goal is to extend the healthy life expectancy of citizens by improving the management rate of chronic diseases through measures such as the expansion of local primary care physicians, the expansion of telemedicine, and the establishment of a public electronic prescription transmission system. The community-based local primary care physician model is planned to be expanded in phases starting in 2026, with the core elements being the provision of comprehensive health management based on multidisciplinary teams and innovative compensation based on performance. The Democratic Party of Korea has submitted a bill to the National Assembly to strengthen primary care and institutionalize the primary care physician system, titled the “Special Act on Strengthening Primary Care” (“Primary Care Special Act”, proposed by Representative In-soon Nam), ahead of the implementation of the Integrated Care Act. The Integrated Care Act and the Primary Care Special Act are expected to serve as the legal and administrative basis for building a society that provides national health care based on primary care. In particular, the National Planning Committee will institutionalize telemedicine based on social consensus and establish a system of telemedicine and remote consultation at public health centers targeting medically underserved areas in rural and fishing villages. Four bills to formally institutionalize telemedicine pilot projects (by Reps. Bo-yoon Choi, Jae-joon Woo, Jin-sook Jeon, and Chil-seung Kwon) are pending in the National Assembly, and the Health and Welfare Committee is expected to review them in the near future. The first subcommittee review is expected to take place as early as the 19th. The National Planning Committee is also expected to include a plan to establish and operate a public electronic prescription transmission system in its national policy task from 2027, based on opinions gathered from the medical community and the Pharmaceutical Association, in line with the institutionalization of telemedicine. The institutionalization of telemedicine and the introduction of public electronic prescriptions were also part of President Jae-myung Lee 's presidential campaign promise. Meanwhile, the National Planning Committee's national policy task must undergo final review by the relevant government ministries and the State Council before it can be finalized.
- Company
- Will Korean companies join in on the sales of obesity drugs?
- by Kim, Jin-Gu Aug 18, 2025 06:02am
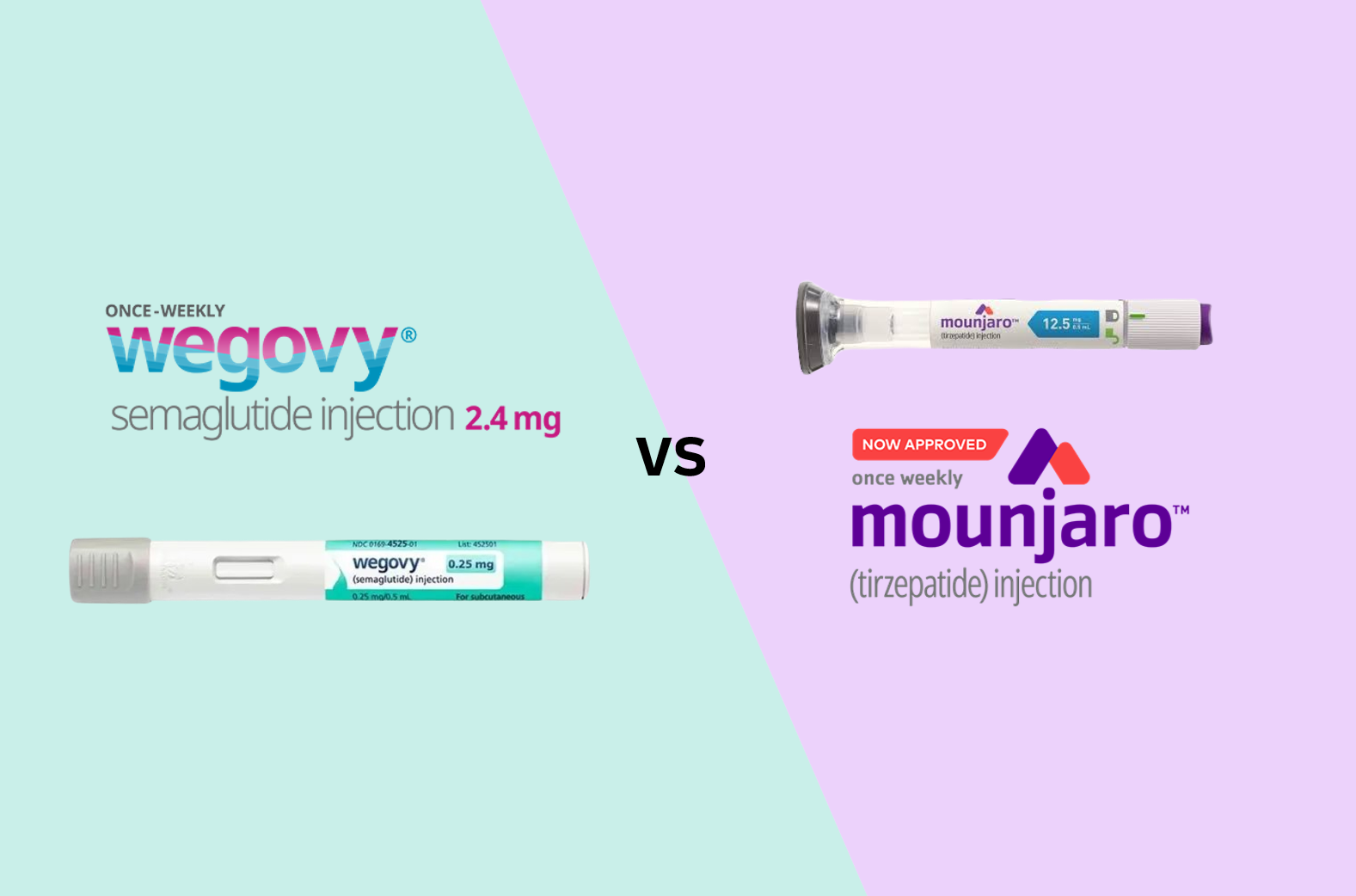
- With the domestic launch of the GLP-1 class obesity treatment Mounjaro (tirzepatide) near, its sales and marketing competition with Wegovy (semaglutide) is already heating up. Lilly has completed preparations for the launch by hiring over 30 additional medical and marketing representatives to handle sales of Mounjaro. Additionally, the possibility of joint sales with domestic pharmaceutical companies is being raised. Novo Nordisk has countered by reducing the domestic supply price of Wegovy by up to 40%. Their discussions on joint sales with Chon Kun Dang are also reportedly ongoing. Mounjaro to be released next week, adds 30 sales staff... Large-scale launch symposium announced According to industry sources on the 14th, Lilly's Mounjaro will be distributed in Korea starting next week. Lilly recently sent a notice to major domestic distributors stating that its supply will begin on the 18th. Hospitals and clinics are expected to receive the product two days later on the 20th. Given the product's record-breaking sales abroad, Lilly Korea is also highly anticipating its launch. In preparation for the launch of Mounjaro, Lilly has hired over 30 additional employees dedicated to sales and marketing for the product. A large-scale launch symposium is also planned. Lilly intends to hold an in-person symposium on the 25th and 26th, followed by an online symposium on the 27th. The company has gone to great lengths to invite Bruno Halpern, the next president of the World Obesity Federation and chief director of the Obesity Centre of Hospital 9 de Julho, São Paulo in Brazil, as a keynote speaker. For now, Lilly Korea plans to directly sell Mounjaro, but the possibility of joint sales with domestic pharmaceutical companies remains open. In the pharmaceutical industry, one major domestic pharmaceutical company is consistently coming up as a potential joint sales partner for Mounjaro. There is speculation that the product may be launched under Lilly directly, then potentially transition to joint sales in the future. The company plans to actively promote Mounjaro's higher weight loss efficacy compared to Wegovy. Mounjaro demonstrated superior weight loss efficacy in the SURMOUNT-5 clinical trial, a head-to-head trial that compared Mounjaro with Wegovy. In the trial, the average weight loss rate at week 72 in the Mounjaro treatment group (10mg or 15mg) was 20.2%, compared to 13.7% in the Wegovy treatment group (1.7mg or 2.4mg), demonstrating a relatively improved weight loss rate. There is also high anticipation in the medical community. A hospital search application is promoting “how to book the lowest-priced hospital for Mounjaro.” Numerous posts promoting advance reservations for Mounjaro are also searchable on major portals and social media platforms. A representative from Lilly Korea stated, “Based on our global experience with the launch of Mounjaro in various countries and our deep understanding of the domestic market accumulated over more than 40 years, we will do our best to help patients with diabetes and obesity in Korea lead better lives with Mounjaro.” Wegovy’s domestic supply price reduced by up to 40% in response...Possibility of joint sales with Chong Kun Dang Ahead of the official launch of Wegovy, Novo Nordisk has responded with a price reduction for Wegovy. Novo Nordisk announced that it will reduce the price of Wegovy by up to 40% starting from the 14th. Excluding the highest dosage of 2.4mg, the prices will be reduced by 40% for 0.25mg, 30% for 0.5mg, 20% for 1.0mg, and 10% for 1.7mg. The price of 0.25mg, which will be reduced by 40%, is expected to be between KRW 200,000 and 220,000. This is analyzed as securing price competitiveness compared to Mounjaro. The supply prices for Mounjaro are ▲2.5 mg KRW 278,066, ▲5 mg KRW 369,307, and ▲7.5 mg and 10 mg KRW 521,377. The 2.5 mg and 5 mg doses will be initially supplied to Korea. The possibility of joint sales with Chong Kun Dang continues to be raised. Its possibility of partnering with Chong Kun Dang for Wegovy’s sales has also been consistently raised. Novo Nordisk and Chong Kun Dang have been in discussions regarding joint sales of Wegovy up until recently, and it is reported that they are nearing a final decision. However, both companies have drawn the line, saying that “nothing has been finalized yet.” If Chong Kun Dang is confirmed as a co-promoter for Wegovy and Mounjaro also signs a contract with a domestic pharmaceutical company, the competitive landscape is expected to expand from a head-to-head battle between two multinational pharmaceutical companies to include domestic pharmaceutical companies as well. The pharmaceutical industry is closely watching how quickly Mounjaro can catch up to Wegovy, which has effectively dominated the market. Wegovy quickly dominated the obesity treatment market after since launch in Korea in October last year. According to pharmaceutical market research firm IQVIA, Wegovy recorded sales of KRW 79.4 billion in the first quarter of this year. Wegovy's market share in the overall obesity drug market reached 73.2%. Wegovy surged to the top of the obesity drug market with sales of KRW 60.3 billion in the fourth quarter of last year, immediately after its launch. Wegovy has also contributed to the expansion of the market size. Prior to its launch in the third quarter of last year, the domestic obesity treatment market size was only KRW 47.4 billion, but it expanded to KRW 93.8 billion in the fourth quarter of last year, nearly doubling in just over three months.
- Company
- "Winrevair, gamechanger for pulmonary arterial hypertension"
- by Whang, byung-woo Aug 18, 2025 06:01am
- Winrevair (sotatercept), a new mechanism treatment for pulmonary arterial hypertension that has emerged after over 20 years, has been approved in Korea. Consequently, competitive market is expected. Although reimbursement remains a challenge, it is expected to change the treatment paradigm for pulmonary hypertension, a disease with significant unmet needs, by addressing the fundamental cause of the disease. Dr. Wook-Jin Chung, President of the Korean Society of Pulmonary HypertensionMSD Korea held a press conference on August 12, celebrating the approval of Winrevair and highlighting the drug's role and value. Pulmonary hypertension is not simply a state of constricted blood vessels. It is a disease of structural narrowing where the walls of the pulmonary arterioles thicken and their lumens become constricted, making it different from general hypertension. Dr. Wook-Jin Chung, President of the Korean Society of Pulmonary Hypertension (Professor of Cardiology at Gachon University Gil Medical Center), explained, "Pulmonary hypertension patients suffer from shortness of breath, cough, fatigue, and fainting as the disease advances. However, due to a complex diagnostic process, patients experience a diagnostic odyssey that can take up to three years." "Pulmonary hypertension patients experience such severe shortness of breath that even walking becomes difficult, leading to serious limitations in all aspects of daily life, including exercise, childcare, housework, and social activities. In severe cases, it can be fatal, with a risk of sudden death." For high-risk patients, the probability of death within one year exceeds 20%, and approximately 30% of domestic patients still die within five years of diagnosis. Until now, phosphodiesterase-5 (PDE5) inhibitors and calcium channel blockers (CCBs) have been used, but they have only been effective in controlling symptoms. Dr. Chung said, "Pulmonary hypertension is both a rare, intractable disease and a chronic condition, so there was an urgent need for a treatment strategy that could restore a healthy daily life to patients." In relation to this, the newly approved Winrevair is generating high expectations because it approaches the fundamental cause of the disease. Winrevair is the first-in-class Activin Signaling Inhibitor (ASI), utilizing the 'Activin' pathway, which is one of the pathogenic mechanisms of pulmonary hypertension. This treatment demonstrates the potential for reverse remodeling by inhibiting the proliferation of abnormally thickened blood vessels, thereby reversing the remodeling of pulmonary arterioles and the right ventricle. In a study of 323 pulmonary hypertension patients, the Winrevair group's 6-minute walk distance (6MWD) increased by 40.1 m, while the placebo group's decreased by 1.4 m. Additionally, Winrevair reduced the risk of clinical worsening or death from all causes related to pulmonary hypertension by 84%. Kyung-hee Kim, Chairperson of the Clinical Guidelines Committee of the Korean Society of Pulmonary Hypertension, said, "Experts from the clinical guidelines committees in Europe and the United States have recommended the addition of ASI when updating pulmonary hypertension treatment guidelines," and added, "The Korean Society of Pulmonary Hypertension is also reviewing the inclusion of ASI in the pulmonary hypertension treatment guidelines in Korea." Dr. Kyung-hee Kim, Chairperson of the Clinical Guidelines Committee of the Korean Society of Pulmonary HypertensionWinrevair was approved through the Ministry of Food and Drug Safety's 'Concurrent Approval-Evaluation-Negotiation Pilot Program' and has quickly entered the reimbursement review process. The Ministry of Health and Welfare (MOHW) has been running the 'Concurrent Approval-Evaluation-Negotiation Pilot Program' since 2023 to improve access to treatment for life-threatening, severe, and rare diseases. The project conducts approval, reimbursement evaluation, and drug price negotiations simultaneously, aiming to shorten the time required for new drugs to be included in the National Health Insurance list. Dr. Chung emphasized, "For patients to benefit from Winrevair, it must be quickly covered by national health insurance." He added, "Even if the treatment is expensive, and its efficacy has been proven overseas, efforts are necessary to ensure it can be used, even if strict criteria are applied."
- Company
- Imfinzi denied reimb for bile duct cancer for the 3rd year
- by Eo, Yun-Ho Aug 14, 2025 06:13am
- Attention is focused on whether there will be progress in the reimbursement process for Imfinzi for bile duct cancer, which has remained non-reimbursed for three years now. According to industry sources, the Health Insurance Review and Assessement Service's Drug Reimbursement Evaluation Committee may likely review the agenda of expanding the reimbursement of AstraZeneca Korea's PD-L1 inhibitor immunotherapy Imfinzi (durvalumab) for bile duct cancer in the second half of the year. In the harsh environment of bile duct cancer, where the average survival period was just over seven months, the emergence of Imfinzi brought about a change in the treatment landscape. Imfinzi demonstrated more than a twofold improvement in overall survival rate compared to the standard chemotherapy regimen at the three-year mark, and in a subgroup analysis of Korean patients, it proved to provide even better survival benefits than the overall patient population. Countries such as Canada, the United Kingdom, Australia, Japan, and Taiwan quickly reimbursed Imfinzi in recognition of its clinical innovation. In the case of the United Kingdom, considering the poor reality of bile duct cancer treatment and the fact that Imfinzi is the first first-line treatment for bile duct cancer, the ICER threshold was applied flexibly by exceptionally weighting the quality-adjusted life years (QALY) during the pharmacoeconomic evaluation process. However, in South Korea, reimbursement barriers remain, limiting patients' access to treatment. It has been three years since Imfinzi was approved by the Ministry of Food and Drug Safety as a first-line treatment for metastatic bile duct cancer. The indication for bile duct cancer was approved by the Cancer Review Committee in November 2024, but there has been no significant progress to date. Jung-yong Hong, Professor of Hematology and Oncology at Samsung Medical Center, said, “The ultimate goal of the government, pharmaceutical companies, and medical professionals is to provide Imfinzi’s treatment benefits, proven as standard therapy, to bile duct cancer patients as quickly as possible. Measures to strengthen institutional flexibility must be explored to enhance treatment access for bile duct cancer patients.”
- Company
- "A paradigm shift in atopic dermatitis treatment"… Adtralza
- by Whang, byung-woo Aug 14, 2025 06:13am
- The treatment landscape is undergoing significant changes as new drugs for atopic dermatitis, including biologics and JAK inhibitors, have emerged over the past few years. Experts assess that patients who gave up on receiving treatments due to recurrence and side effects are revisiting hospitals with the introduction of new drugs. As treatment options become diverse, the primary concern for the clinical field is 'what' treatment to use. During the meeting with DailyPharm, Professor Seong Jun Seo of Soon Chun Hyang University Cheonan Hospital's Department of Dermatology emphasized the importance of personalized treatment tailored to each patient, along with consideration of economic factors. More treatment options to treating atopic dermatitis…"personalized treatment is key" Atopic dermatitis treatment options have expanded beyond the previous standard therapies, which include moisturizers and topical steroids, to two types: biologics and oral JAK inhibitors. Currently, three biologics and a JAK inhibitor are listed for reimbursement in Korea. Professor Seong Jun Seo of Soon Chun Hyang University Cheonan HospitalProfessor Seo advises that personalized treatment, based on patient characteristics, is key as treatment options continue to expand. Professor Seo said, "In my opinion, criteria for selecting a treatment should be based on the patient's condition and needs, and that 'the best drug should be the first one used'," and stressed, "It is crucial to provide the best treatment option for each patient from the very beginning." For example, for patients with atopic dermatitis who also have other allergic conditions (such as asthma or rhinitis), or those who are elderly or have chronic diseases, a biologic with a lower burden on the systemic immune system is considered first. In contrast, for patients who desire a quick effect or have a strong fear of injections, an oral JAK inhibitor is used first. This means that a personalized treatment, which considers both treatment goals and patient preferences, is key to treatment success. However, in reality, insurance criteria are a stumbling block. Under the current National Health Insurance's Special Case Medical Expense Coverage criteria, new drug treatment is limited to severe patients (EASI score ≥23, BSA ≥10%). This leaves moderate patients in a reimbursement blind spot, and experts unanimously agree that this needs to be improved to widely support personalized treatment. Professor Seo also pointed out that "more flexible criteria are needed, such as giving weighted scores to lesions on areas that significantly affect quality of life, like the face and neck, even if the EASI score is below 23." One Year After Adtralza's Launch…Patients are more satisfied with improved safety and facial flusing In this situation, the evaluation from the field is that Adtralza (tralokinumab) from LEO Pharma, which was the second biologic to be listed in Korea, is gaining favor with many patients since its launch last year. Adtralza, which has an IL-13 single-target mechanism that selectively targets only interleukin-13, was noted as a strong competitor to the existing Dupixent (which has an IL-4/13 dual-target mechanism). In clinical trials, Adtralza achieved an EASI-75 score in 56% of patients at week 16, demonstrating its superior efficacy compared to placebo. It also showed a 38.9% achievement rate for an IGA score of 0/1 (almost clear of lesions). Adtralza was listed for health insurance reimbursement in May 2024. In its first year, it has successfully established itself in the prescription lists of university hospitals, rapidly expanding its market share. What are Adtralza's unique strengths, then? Professor Seo, who has prescribed various treatments in the clinical setting, highlighted Adtralza's excellent safety profile. Professor Seo explained, "Dupixent generally has high satisfaction due to its excellent efficacy and low side effects, but its drawbacks include unexpected side effects like conjunctivitis and facial flushing," and added, "While Adtralza can also cause conjunctivitis, the incidence is lower than with Dupixent, and it shows an effect in improving symptoms in patients who experienced facial flushing with Dupixent." As improvement of symptoms in exposed areas is directly related to a patient's quality of life, Adtralza could be a favorable option to reduce the disease burden for atopic dermatitis patients who experience social avoidance due to severe inflammation on their face or neck. Patient A, who was present with Professor Seo, said, "After being treated with Adtralza, my symptoms improved noticeably over time. I took it for about half a year after the initial dose and felt a clear treatment effect." Another strength is its cost-effectiveness. Regarding this, Professor Seo said, "Adtralza's drug price is lower than that of other biologics. It's about 60% of Dupixent's cost, which makes it a preferred option for patients with a significant financial burden." Given the long-term nature of atopic dermatitis treatment, the cost of medication is a concern for both patients and medical professionals. In terms of cost-utility, Adtralza's superior accessibility is notable. Limitations of inter-class switching…highlighted biomarker importance With the increasing number of new treatment options, the project of creating an environment that benefits patients is gaining prominence. This year, in response to requests from the field, the government partially relaxed reimbursement criteria to allow inter-class switching between biologics and JAK inhibitors. However, the lack of a provision for changing within the same class remains a point of regret. Professor Seo said, "Atopic dermatitis is a complex disease with various contributing factors, so a more flexible insurance application is needed for treatment selection," and added, "The symptom patterns and patient responses can vary, so when one drug is insufficient, a path should be opened to allow switching to a different drug within the same class." Lastly, Professor Seo emphasized the importance of biomarker development for the future of atopic dermatitis treatment. He explained that although symptoms can now be suppressed for long periods with available therapies, personalized indicators are necessary to establish precision medicine. Professor Seo added, "Atopic dermatitis is a difficult disease that requires long-term management, but with recent treatment innovations, it is becoming a manageable chronic disease." He concluded, "To implement precision medicine, we need personalized indicators for each patient, and when predictive treatment technologies are introduced in the future, we can take another step closer to curing atopic dermatitis."
- Company
- Only Vemlidy’s sales grew in the ₩300B HBV drug mkt
- by Kim, Jin-Gu Aug 14, 2025 06:13am
- Korea's hepatitis B treatment market, which is valued at KRW 300 billion annually, is rapidly restructuring around Vemlidy (tenofovir alafenamide). Among original products, only Vemlidy is maintaining its growth, and in the generic market, only the Vemlidy series has shown a significant increase in sales. HBV treatment market reaches KRW 150.9 billion in 1H, up 2% year-on-year According to the pharmaceutical market research institution UBIST on the 13th, the domestic hepatitis B treatment market reached KRW 150.9 billion in the first half of this year. This represents a 2% increase from the KRW 147.6 billion in the first half of last year. At this pace, the HBV drug market is expected to record prescription sales of over KRW 300 billion for the second consecutive year. Share of Vemlidy original and generic products in the Quarterly HBV treatment market Last year, the hepatitis B treatment market re-entered the KRW 300 billion range for the first time in 6 years since 2018. The market had exceeded KRW 300 billion in 2018 but decreased to KRW 273.1 billion in 2019. This was due to the expiration of the patent for Gilead Sciences' Viread, which had been leading the market, and the subsequent price reduction of the drug. The market further shrank to KRW 266 billion in 2020. Since 2021, the market has shifted to growth. In 2021, the market increased by 4% to KRW 275.6 billion. In 2022, it reached KRW 283.8 billion, then grew by 3% for two consecutive years to KRW 283.8 billion in 2022, then KRW 292.3 billion in 2023. Last year, it expanded further to over KRW 300 billion. Vemlidy becomes the only original drug to show growth…grows 10% in one year Gilead's Vemlidy drove market growth. Vemlidy’s first-half prescription sales reached KRW 37.8 billion, a 10% increase from the previous year. Vemlidy is a new hepatitis B drug developed by Gilead as a successor to Viread. While the existing Viread is highly effective in suppressing the hepatitis B virus, its drawbacks include side effects such as renal dysfunction and reduced bone density. Vemlidy overcomes these limitations of Viread. In clinical trials, no such side effects were observed. Given the challenging nature of hepatitis B, which is difficult to cure, the long-term safety of Vemlidy has emerged as a key advantage. Quarterly prescriptions of major HBV treatments (Viread, Baraclude, Vemlidy) Launched in Korea in 2017, Vemlidy is the only original product among major products that has maintained growth. Meanwhile, the market leader, Viread (tenofovir sofosbuvir), saw a 1% decrease from KRW 45.4 billion to KRW 44.9 billion in one year. Sales of BMS's ‘Baraclude (entecavir)’ decreased by 1% from KRW 35.3 billion to KRW 35.1 billion. In addition, ‘Zeffix (lamivudine)’, ‘Besivo (besifovir)’, ‘Levovir (clevudine)’, and ‘Sebivo (telbivudine)’ all saw a decrease in prescriptions. Hepsera (adefovir) has been completely withdrawn from the market since 2023. Among the major products, while Viread and Baraclude saw a slowdown, Vemlidy was the only one to show an increase in its prescription sales, leading to changes in the market rankings. Vemlidy ranked third in the market behind Viread and Baraclude until the first half of last year, but then surpassed Baraclude to move up to second place. The gap with Viread narrowed from KRW 11 billion to KRW 7.2 billion in just one year. Generic market also sees rapid growth of Vemlidy follow-on drugs... accelerates market restructuring The same trend has been observed in the generic market. Vemlidy generics are the only ones that showed continued growth. The combined prescription amount for Vemlidy generics in the first half of this year was KRW 4.1 billion, more than double the KRW 2 billion recorded in the same period last year. Although it showed a somewhat sluggish trend initially after its official launch in February 2023, its growth has accelerated since last year. Sales of Samil Pharmaceutical's ‘Vemlino’ increased from KRW 9 billion in the first half of last year to KRW 18 billion in the first half of this year, while sales of Dong-A ST's ‘Vemlia’ increased from KRW 7 billion to KRW 13 billion, each more than doubling in sales. The remaining products also saw their prescription sales increase by more than twofold. On the other hand, Baraclude generics and Viread generics have seen a slowdown in their upward trend. The combined prescription sales of Baraclude generics decreased by 3% from KRW 16.3 billion to KRW 15.8 billion over the past year. Sales of Viread generics also decreased by 3% from KRW 7.2 billion to KRW 7 billion. The industry expects that the market restructuring centered on Vemlidy will accelerate in both the original and generic markets. This is because Bemliride has already been recommended as a first-line treatment by liver associations in the US, Europe, and South Korea. In South Korea, its reimbursement has been approved for the treatment of non-cirrhotic liver cirrhosis and liver cancer since 2022.
- Policy
- Gilead’s Yescarta approved in KOR...3rd CAR-T therapy
- by Lee, Hye-Kyung Aug 14, 2025 06:13am
- The third CAR-T therapy has been approved in Korea. The Ministry of Food and Drug Safety approved Gilead Sciences Korea's Yescarta (axicabtagene ciloleucel) today (13th). Yescarta is a CAR-T therapy that received approval from the U.S. FDA in 2017 and the European EMA in 2018. Last year, the EMA approved it as a second-line treatment for patients with adult diffuse large B-cell lymphoma and B-cell acute lymphoblastic leukemia. In June, the FDA expanded the indication for Yescarta to include patients with follicular lymphoma. In Korea, after the drug was designated as an orphan drug by the MFDS in September last year, it has been undergoing the formal approval process. According to a brief issued by the Korea Biotechnology Organization, Yescarta is currently the CAR-T therapy with the highest market share worldwide. Posting sales of USD 1.5 billion (approximately KRW 2 trillion) last year, Yescarta ranked first with a 40% share of the entire CAR-T market. It was followed by Novartis' Kymriah posting USD 500 million (approximately KRW 700 billion), Johnson & Johnson's ‘Carvykti’ at USD 500 million, BMS's ‘Abecma’ at USD 470 million (approximately KRW 650 billion), and Gilead Sciences' ‘Tecartus’ at USD 370 million (approximately KRW 500 billion). Kymriah and Carvykti have already been granted marketing authorization in Korea. Meanwhile, according to an analysis presented at the American Society of Clinical Oncology (ASCO) 2023 Annual Meeting, at a median follow-up of 47.2 months, the overall survival (OS) in the Yescarta treatment group had not yet reached the median, while the placebo group had a median OS of 31.1 months, indicating a statistically significant 27% lower risk of death with Yescarta. The estimated 48-month overall survival rate was 54.6% for Yescarta and 46.0% for the control group, with Yescarta demonstrating consistent survival benefits across pre-specified subgroups, including age, primary resistance, early relapse, and high-grade B-cell lymphoma.
- Company
- 'Tevimbra' applies for reimbursement for five indications
- by Eo, Yun-Ho Aug 13, 2025 06:07am
- Product photo of Tevimbra 'Tevimbra' is quickly expanding following its successful inclusion in the insurance reimbursement for esophageal cancer. BeOneMedicines Korea has been confirmed to have applied for reimbursement on the day of approval for five new indications for its PD-1 inhibitor immunotherapy Tevimbra (tislelizumab), which received additional approval in June. In April, Tevimbra became the first immunotherapy to receive reimbursement for esophageal cancer. Following this, it has added five new indications for solid tumors, including esophageal cancer, gastric cancer, and non-small cell lung cancer. The specific expanded indications are ▲first-line combination therapy for unresectable, locally advanced, or metastatic esophageal cancer ▲first-line combination therapy for unresectable or metastatic, HER2-negative gastric or gastroesophageal junction adenocarcinoma ▲two types of first-line combination therapy, and second-line monotherapy for non-small cell lung cancer. Tevimbra has already received approval from the U.S. FDA for first- and second-line treatment of unresectable or metastatic esophageal squamous cell carcinoma, as well as for first-line treatment of advanced gastric cancer. The European Medicines Agency (EMA) has also approved it for first- and second-line treatment of esophageal squamous cell carcinoma, first-line treatment of advanced gastric cancer, and first- and second-line treatment of non-small cell lung cancer. Tevimbra is receiving various expanded indication in the global market. Since the company, BeOne Medicines, has been quick to proceed with reimbursement procedures for additional indications, Tevimbra's role is expected to expand across various cancer types in Korea. Expectations are particularly high due to BeOne Medicines' previous track record of successfully concluding negotiations with the government by advocating for 'reasonable drug pricing' since its initial listing. The reason for the low reimbursement rate by indication for immunotherapies, including esophageal cancer, is, as expected, related to drug pricing and financial burden. After reimbursement was applied to some cancer types, like lung cancer, the total amount claimed for immunotherapies and their share of the total cancer drug claims within the health insurance system significantly increased, raising concerns about the financial burden. As of 2023, cancer drug claims totaled KRW 2.4 trillion, with immunotherapy claims accounting for approximately KRW 500 billion, or 20% of the total cancer drug claims. It remains to be seen whether BeOne Medicines can uphold its company philosophy of 'providing innovative new drugs at a reasonable price and leaving no patient behind.' Meanwhile, the efficacy and safety of Tevimbra were proven in various indications through the RATIONALE clinical trial series (RATIONALE-303, 304, 305, 306, 307). In esophageal squamous cell carcinoma and gastric or gastroesophageal junction adenocarcinoma, Tevimbra demonstrated clinical benefits in the overall patient population. The trials showed consistent results in pre-specified subgroups based on PD-L1 expression.
- Policy
- New drug approval fees ₩410mil in KOR
- by Lee, Hye-Kyung Aug 13, 2025 06:07am
- Since the new drug approval fee was significantly increased to KRW 410 million starting this year, a total of 14 new products containing 10 ingredients have been submitted for approval. Although the specific product names cannot be disclosed, dedicated teams have been formed for 6 chemical drug substances and four biopharmaceutical substances, and the review process is currently underway. On the 12th, Young-joo Kim, Director of the Drug Approval Division at the Ministry of Food and Drug Safety, So-hee Kim, Director of the Director of the Cardiovascular Drug Division of the Drug Review Department, and Jae-ok Kim, Director of the Biological Products Division of the Bio and Herbal Medicine Review Department, met with specialized media journalists to provide an interim briefing on the new drug approval and review innovation process that began this year. , Young-joo Kim, Director of the Drug Approval Division at the Ministry of Food and Drug Safety, So-hee Kim, Director of the Director of the Cardiovascular Drug Division of the Drug Review Department, and Jae-ok Kim, Director of the Biological Products Division of the Bio and Herbal Medicine Review Department On January 1, the MFDS raised the new drug approval fee to KRW 41 million, formed dedicated teams for each item, expanded face-to-face consultations and reviews between companies and approval reviewers for items submitted for approval to a maximum of 10 times, and shortened the manufacturing and quality control evaluation and actual condition survey of new drug manufacturing facilities (to within 90 days). The increased approval fees were to be used mostly to hire high competency reviewers, and the MFDS has established a policy to operate a professional, swift, transparent, and predictable approval review system so that the process from new drug approval application to license issuance can be completed within 295 days. The new drug approval and review process begins with a pre-submission consultation, followed by the submission of a product license application. A dedicated team of approximately 15 members is then formed for each product. Actual meetings begin within 2 weeks of the submission of the application. GMP inspections for new drugs are also completed within 90 days of the submission date. On January 31, multinational pharmaceutical company Eli Lilly applied for marketing authorization for its new breast cancer drug “Inluriyo Tab (imlunestrant),” and a total of 10 substances are currently under review. While actual marketing authorizations are not completed in the order of application submission, for Inluriyo, if the innovative approval scheme for new drugs is applied, approval could be granted as early as November or by December at the latest. Director Young-Joo Kim stated, “While I cannot comment on the approval process for specific products, we are striving to complete the approval process within 295 days from the date of submission of the marketing authorization application in accordance with the new drug approval and review procedures. Despite the increase in new drug approval fees, we anticipate that the number of marketing authorization applications submitted will be similar to previous years.” As of August, applications for new drug approvals for 10 ingredients have been submitted, and based on the usual number of applications, applications for 20 ingredients were submitted in 2023 and 19 ingredients in 2024. The MFDS believes that if this trend continues, the number of item approval applications will remain similar to those of previous years. Kim added, “Until last year, when requests for supplementary data were made, everything was handled via documents. However, now, there are over 10 face-to-face meetings from preliminary consultations to supplementary data submission requests, and all of this process is documented.” However, despite the significant increase in fees, only 15 to 17 dedicated staff members are being assigned to expedite approvals. This is why, when a meeting is held regarding supplementary materials, everyone has to gather for a meeting lasting over an hour and a half. Director Jae-ok Kim explained, “One hour and a half may seem short, but when supplementary materials are submitted, pharmaceutical companies also select the points they want to focus on and ask questions. The meeting lasts about an hour and a half, with the purpose of the supplementary data request being explained and any questions being answered.” In the case of new drug approval fees, most of the increased amount will be used to cover the pay of high competency reviewers. Currently, there are 31 high competency reviewers hired specifically for the new drug team, which is close to the total quota of 36. The new drug dedicated team is composed of a team leader who is the head of the approval department, a product manager from the approval department, and specialists in safety and efficacy, quality control, GMP (good manufacturing practice), and GCP (good clinical practice), rendering the need to reinforce the specialized review staff. Director Young-joo Kim said, “With the increase in new drug approval fees, the cost of review is borne by the beneficiaries. The MFDS has a total of 370 reviewers, including those for medical devices, and about 10% of them are high competency reviewers.” However, as high competency reviewers are hired as public employees rather than civil servants, it is difficult for them to provide stable service. As a result, they may only receive training on high competency review techniques and move to other organizations within a few years. Director Jae-ok Kim added, “The current review staff is going through a transitional period. High competency reviewers cannot be immediately deployed; they must first undergo training, work as assistant reviewers, and then transition to head reviewers." He also pointed out that “this process is causing an overload of work for existing review staff.” Director So Hee Kim also emphasized, “Since the system is still in its initial stages, mid-level managers are facing significant challenges. While hiring high-skilled reviewers is important, expanding the pool of personnel capable of fulfilling mid-level management roles is also necessary.” In this regard, Director Young-joo Kim said, “It is important to enable high competency reviewers hired as public officials to be appointed civil servants,” adding, “We plan to consult with other ministries and request that the number of civil servants be continuously increased.” Although the MFDS is applying the new drug approval fee hike only to new drugs, it plans to gradually expand it to other items. President Jae-myung Lee's remarks also support this stance. At a recent cabinet meeting, the president stated, “Insufficient review costs and a shortage of personnel are causing delays in new drug approvals, which is a loss for society as a whole. Increasing review costs is a way to expand the budget without increasing the financial burden.” Director Young Joo Kim said, “The MFDS understands that the intention is not to further raise the new drug approval fees, but to expand the fees to items other than new drugs to increase revenue. We are in ongoing discussions with government ministries and related industries on items for which fees can be raised based on principles such as the novelty and complexity of the substance.”
- Product
- Mounjaro set to hit shelves next week... pricing a dilemma
- by Jung, Heung-Jun Aug 13, 2025 06:07am
- With the imminent arrival of the diabetes and obesity treatment Mounjaro (tirzepatide) in Korea, pharmacies are struggling to set an appropriate price for the item. Due to the nature of high-priced non-reimbursable drugs, there are regional price differences, and price competition is expected in the future, so pharmacies are showing a cautious stance on their initial sales price. Recently, some distributors notified pharmacies of Mounjaro ‘s supply schedule for the 20th and surveyed the pharmacies’ willingness to handle the product to anticipate demand. Additionally, telemedicine platforms are guiding affiliated clinics and pharmacies to input their planned selling prices. Like Wegovy, Mounjaro’s supply price was disclosed before its domestic launch. However, unlike Wegovy, the supply price varies by dosage. While there may be minor differences depending on the distributor, the prices are currently set at KRW 278,000 for 2.5mg and KRW 369,300 for 5mg. The prices for the upcoming 7.5mg and 10mg doses are reported to be KRW 521,300. Pharmacists are expected to monitor the prices set by nearby clinics and pharmacies before determining their own sales prices. Pharmacist A from Seoul stated, “Based on the 2.5mg dosage, the price is expected to range from the mid-300,000 KRW to the 400,000 KRW bracket. Although distribution is scheduled for next week, we have not yet decided whether to carry the product, as we are unsure whether hospitals will issue outpatient prescriptions.” Pharmacist A added, “Although both products are used for obesity treatment, their mechanisms of action are different, so it is not appropriate to simply compare their dosage and price.” In the case of Wegovy, the selling price, which was in the range of KRW 500,000 to KRW 600,000 when it was first launched in Korea, gradually decreased, and the price was adjusted. In the case of obesity treatments, price information is quickly shared through telemedicine platforms and communities, so even among non-reimbursable drugs, the sales price can fluctuate greatly. Some pharmacists are hesitant to handle the drug because the supply price is known and there is a risk that it could become dead stock. Pharmacist B from Seoul stated, “It has a competitor, so it's difficult to predict how much demand there will be for Mounjaro, so we're waiting to see how demand develops. We also have remaining stock of Saxenda and Wegovy. Price competition is intense, so expanding our inventory can be a burden.”