- LOGIN
- MemberShip
- 2026-04-03 19:59:15
- Policy
- MFDS's bill on stable supply of essential drugs in review
- by Lee, Jeong-Hwan Aug 20, 2025 06:22am
- A bill to add “medicines that are essential for healthcare and require stable supply” and “medicines with similar therapeutic effects with no alternative treatments” to the list of national essential medicines, thereby resolving the issue of unstable supply of medicines, is likely to pass the Health and Welfare Committee's legislative subcommittee. At the subcommittee meeting held on the morning of the 19th, the subcommittee members and the Ministry of Food and Drug Safety agreed on the intent of the legislation, but there were some disagreements over the specific wording that should be used to codify the definition, so the subcommittee agreed to pass the bill after going through the amendment process on the same day. The subcommittee members and the MFDS also agreed to accept a provision to revise the designation method for essential medicines to be designated by the Minister of Health and Welfare and the Minister of Food and Drug Safety after consultation with the National Essential Medicine Stable Supply Council. The bill also includes provisions to include “linking drug distribution information” in the scope of the Comprehensive Drug Management Center's duties and to allow the MFDS director to request the Drug Management Information Center to provide and link drug distribution information. On the morning of the same day, the subcommittee members decided to merge the bills proposed by Representative Sun-min Kim of the People Power Party and Representative Mi-hwa Seo of the Democratic Party of Korea and continue deliberations. However, as further discussions on the bill are scheduled at the afternoon subcommittee meeting, there is a possibility that it could pass the subcommittee on the same day if an agreement is reached between the government and the subcommittee members. The bill expands and revises the definition and scope of essential medicines and establishes a legal basis for managing distribution information to contribute to the stable supply of essential medicines. Expanding the definition of essential medicines to strengthen management of drugs with unstable supply The current law defines essential medicine as “medicines that are essential for health and medical care, such as disease control and radiation disaster prevention, but whose stable supply is difficult to ensure through market functions alone, and which are designated by the Minister of Health and Welfare and the Minister of Food and Drug Safety in consultation with the heads of relevant central administrative agencies.” Rep. Sun-min Kim's bill proposed revising the definition of essential medicines to “drugs that are essential for maintaining the national health system, such as disease control and radiation disaster prevention, or that are essential for health and medical care and require a stable supply, as designated by the Minister of Health and Welfare and the Minister of Food and Drug Safety in consultation with the National Essential Medicine Stable Supply Council.” Representative Mi-hwa Seo's bill proposed amending the definition to “medicines that are essential for health and medical care, such as disease control and radiation disaster prevention, but whose stable supply is difficult to ensure through market functions alone, or drugs for which there are no substitutes with similar therapeutic effects, as designated by the Minister of Health and Welfare and the Minister of Food and Drug Safety after consultation with the National Essential Medicine Stable Supply Council pursuant to Article 83-4, Paragraph 3.” The MFDS agreed to reflect both Kim and Seo’s bills and submitted a proposed amendment to the relevant clause. The MFDS proposed to revise the bill so that essential medicines would be designated by the Minister of Health and Welfare and the Minister of Food and Drug Safety after consultation with the National Essential Medicine Stable Supply Council, and to define essential medicine as drugs that are essential for maintaining the national health system, drugs that are essential for health care but difficult to supply stably through market forces alone, and drugs for which there are no substitutes with similar therapeutic effects. The members of the subcommittee raised the need to define essential medicines in one line rather than listing them individually as proposed in the MFDS amendment, and requested a further amendment. The subcommittee is expected to resume its review of the bill in the afternoon based on the amendment. Distribution information linkage included in the scope of the Integrated Pharmaceutical Management Information Center’s duties The MFDS agreed to add “distribution information linkage” to the scope of the Integrated Pharmaceutical Management Information Center’s duties and to allow the MFDS Minister to request the provision and linkage of distribution information in consultation with the director of the information center. The MFDS also accepted provisions to add “drugs that are not designated as essential medicines but require stable supply due to temporary increases in demand” to the scope of drugs managed by the National Essential Medicine Stable Supply Council and to add monitoring of essential drug supply and demand trends to the duties held by the Minister of Health and Welfare and the Minister of Food and Drug Safety. The MFDS also expressed its opinion regarding the provision to specify the composition of the National Essential Medicine Stable Supply Council in the law. Under the current law, the council is composed of 20 government members, including one chairperson, with the remaining details to be specified in the implementing regulations. Rep. Sun-min Kim’s proposal stipulates that the council shall consist of 30 members, including two chairpersons, with government and private-sector members, and that private-sector members shall constitute a majority. Rep. Mi-hwa Seo's proposal stipulates that the council shall consist of 30 members, including one chairperson, with government and private-sector members. The MFDS proposed a council composition similar to Rep Kim’s proposal, consisting of 30 members, including two chairpersons, but added patient group representatives as recommended by Rep Seo. The subcommittee plans to resume deliberations in the afternoon, as it believes that there has not been sufficient discussion on the details of the bill.
- InterView
- Dong-A ST launches Sterlara biosimilar Imuldosa in the US
- by Cha, Jihyun Aug 20, 2025 06:22am
- Dong-A ST (CEO: Jae-Hoon Jeong) announced on the 19th that the company has launched IMULDOSA (project name DMB-3115, active ingredient ustekinumab), a biosimilar version of Stelara, in the US through its partners, multinational pharmaceutical companies Intas Pharmaceuticals and Accord Biopharma. IMULDOSA is biosimilar version of Janssen’s Stelara that is indicated for the treatment of inflammatory conditions such as plaque psoriasis, psoriatic arthritis, Crohn's disease, and ulcerative colitis. Stelara has generated global sales of approximately USD 21.552 billion (IQVIA 2024 cumulative revenue). IMULDOSA has been launched in 14 countries, including Germany, the United Kingdom, and Spain, and has obtained marketing authorization in the MENA region, including Saudi Arabia, Qatar, and the United Arab Emirates. A representative from Dong-A ST stated, “With the launch of IMULDOSA in the United States following its introduction in Europe, our global market expansion is gaining momentum. We will strive to ensure that IMULDOSA becomes an effective treatment option for patients worldwide.” IMULDOSA was jointly developed by Dong-A Socio Holdings and Meiji Seika Pharma since 2013. In July 2020, the rights for development and commercialization were transferred to Dong-A ST to facilitate efficient global project execution, and the two companies have since continued joint development. In July 2021, a global license-out agreement for IMULDOSA was signed with the multinational pharmaceutical company Intas Pharmaceuticals. Intas is commercializing IMULDOSA through its global subsidiaries, including Accord BioPharma in the U.S. and Accord Healthcare in Europe, the UK, and Canada.
- Company
- Dapa N with Forxiga's indication leads the mkt by surprise
- by Kim, Jin-Gu Aug 20, 2025 06:22am
- Product photo of Forxiga and Dapa N Prescription sales of HK inno.N's 'Dapa N,' which received transfer of Forxiga indications, have surged in the SGLT-2 inhibitor diabetes treatment market. It has risen to the top of the dapagliflozin monotherapy market. Analysis suggests that this success is attributed to Dapa N's transition in indications, including chronic heart failure, following the withdrawal of the former market leader, Forxiga, from the Korean market. With the reimbursement scope for chronic kidney disease recently expanded in the second half of the year, Dapa N's upward trend is expected to continue for some time. HK inno.N's Dapa N Prescription Sales Jump from KRW 300 Million → KRW 2.5 Billion in a Year According to the pharmaceutical market research firm UBIST on August 18, HK inno.N's Dapa N recorded prescription sales of KRW 2.5 billion in Q2, a significant increase from KRW 300 million in the same period last year. Dapa N showed sluggish performance in its early launch, with sales remaining below KRW 500 million for six consecutive quarters after its release in Q2 of 2023. Notably, even after Forxiga's indications were transferred to Dapa N in April of last year, it did not immediately lead to an increase in prescription sales. At that time, AstraZeneca Korea withdrew Forxiga's product approval while simultaneously granting clinical data, thereby transferring Forxiga's indications to Dapa N. Quarterly Prescription Sales Performance by Key Dapagliflozin Monotherapies (unit: KRW 100 million, source: UBIST) Index: RED-HK inno.N However, an analysis shows that Dapa N's growth has become steep since Q4 of last year. Its prescription sales, which were below KRW 500 million, surged to KRW 1.2 billion in Q4 of last year, KRW 2.3 billion in Q1 of this year, and KRW 2.5 billion in Q2. This is analyzed as a shift in the focus of prescriptions towards Dapa N, as the distribution volume of Forxiga gradually decreased in Korea. After deciding to withdraw Forxiga from Korea at the end of 2023, AstraZeneca Korea halted the supply of new inventory in the second half of last year, only distributing existing stock domestically. Dapa N Leads After Forxiga's Departure… Will Upward Trend Continue with Expanded Reimbursement? During this process, Dapa N became the leader in the dapagliflozin monotherapy market. In Q1, with Forxiga completely withdrawn, Dapa N recorded KRW 2.3 billion in prescription sales, surpassing Boryung's Trudapa (KRW 1.3 billion) to take the lead. In Q2, it further widened the gap with Trudapa. The pharmaceutical industry expects Dapa N's growth momentum to continue for some time, as its reimbursement scope was expanded to include chronic kidney disease last month. As of July 1, the government expanded Dapa N's reimbursement to patients with non-diabetic chronic kidney disease. The conditions for reimbursement are: concurrent administration with other standard kidney disease treatments for patients ▲Who have been stably treated with an ACE inhibitor or Angiotensin II receptor blocker at the maximum tolerated dose for at least 4 weeks ▲Who have an eGFR of 20–75ml/min/1.73㎡ ▲Who have a positive urine dipstick test (1+ or more) or a uACR of 200mg/g or more. The key factor is the performance of other dapagliflozin monotherapies. The fact that the reimbursement scope for 15 products from 9 pharmaceutical companies has become identical to Dapa N's since last month is expected to be a significant factor. Previously, Dapa N was the only one to hold indications for chronic heart failure and chronic kidney disease, in addition to diabetes. Starting last month, products from 9 companies, including ▲Boryung's Trudapa ▲Hanmi Pharmaceutical's Daparon ▲KyungDong Pharmaceutical's Dapajin ▲Chong Kun Dang's Exiglu ▲Daewon Pharmaceutical's Dapawon ▲Dong-A ST's Dapapro ▲Samik Pharmaceutical's Difaglu ▲Daewoong Bio's Forxidapa ▲Nex Pharm Korea's Floga, now have the same indications as Dapa N. SGLT-2i Monotherapy Market Has Expanded 3%...Jardiance 25%↑·Envlo 16%↑ The SGLT-2 inhibitor monotherapy market, including Dapa N, grew by 3% from KRW 40.5 billion in Q2 of last year to KRW 41.8 billion in Q2 of this year. Boehringer Ingelheim's Jardiance (empagliflozin) and Daewoong Pharmaceutical's Envlo (inavogliflozin) performed well. Jardiance's prescription sales increased by 25% over one year, rising from KRW 16.1 billion to KRW 20.1 billion. Envlo's sales increased by 16% in the same period, from KRW 2.5 billion to KRW 2.9 billion. It appears that the market gap left by Forxiga has been filled by Jardiance, Envlo, and Forxiga's generics. Quarterly Prescription Sales Performance by Key Dapagliflozin Monotherapies (unit: KRW 100 million, source: UBIST) Index: BLUE-AstraZeneca This market is expected to shift again in October of this year when Jardiance's substance patent expires. Currently, around 50 pharmaceutical companies have received approval for Jardiance generics and are ready for launch. The industry anticipates a repeat of the fierce generic competition in the diabetes treatment market.
- Company
- Reimb for 'Padcev' combo as a 1st-line treatment reapplied
- by Whang, byung-woo Aug 19, 2025 06:12am
- Will 'Padcev+Keytruda' combination therapy overcome the reimbursement hurdle in the area of metastatic urothelial cancer, where the first-line treatment option had not been available? In terms of treatment effectiveness, there is no disagreement among experts that, in the long term, it is a first-line standard treatment option. With Astellas now taking proactive steps, reimbursement entry will ultimately be the key factor. Astellas Pharma Korea held a press conference on August 18 to celebrate the first anniversary of the domestic approval of Padcev (enfortumab vedotin) in combination therapy for first-line metastatic urothelial carcinoma Astellas Pharma Korea held a press conference on August 18 to celebrate the first anniversary of the domestic approval of Padcev (enfortumab vedotin) in combination therapy for first-line metastatic urothelial carcinoma and shared its future strategy. The Padcev + Keytruda combination therapy was approved by the Ministry of Food and Drug Safety (MFDS) last July for the first-line treatment of locally advanced or metastatic urothelial carcinoma. The basis for the approval was the Phase 3 KEYNOTE-A39/EV-302 clinical trial. The results, which evaluated the efficacy of the Padcev + Keytruda combination therapy in patients with previously untreated locally advanced or metastatic urothelial carcinoma, showed that it approximately doubled both the overall survival (OS, 31.5 months) and the progression-free survival (PFS, 12.5 months) compared to platinum-based chemotherapy. Professor Jeong Min Cho of the Department of Oncology at Ewha Womans University Mokdong Hospital explained, "Metastatic urothelial carcinoma is a type of cancer with a very low survival rate of 14.3%, giving it a poor prognosis similar to highly fatal lung cancer." She added, "Due to its aggressive nature and the high proportion of elderly patients, the patient's condition often deteriorates rapidly during the early stages of treatment." Professor Jeong Min Cho of the Department of Oncology at Ewha Womans University Mokdong Hospital Professor Cho continued, "The Padcev first-line combination therapy has been confirmed to show therapeutic effects similar to those in clinical studies in clinical practice, establishing itself as an option that can provide a powerful treatment response in the early stages of treatment." A year after its approval, the demand from the field for the Padcev + Keytruda combination therapy is for expanded access through reimbursement. On this day, Professor Cho said, "In clinical practice, we are constantly getting inquiries from patients about whether it's covered by insurance." In addition to the drug's efficacy, hurdles such as the cost burden create apparent limitations in treatment access. Professor Cho emphasized, "It is often difficult to recommend to patients due to the high cost burden, and I feel heavy-hearted that we cannot provide the best treatment opportunity on time." She stressed, "As Padcev is recommended as the top first-line option in major global guidelines, institutional support is urgently needed in Korea as well." Currently, Astellas has reportedly re-applied to the health authorities for reimbursement for both the Padcev + Keytruda combination therapy and Padcev monotherapy for first-line and later treatment of metastatic urothelial carcinoma. It is also known to have proactively submitted a proposal for sharing financial burdens. This is interpreted as a move to secure reimbursement for both combination therapy and monotherapy simultaneously, in preparation for submission to the upcoming Cancer Disease Review Committee in September. However, a view exists that a company's strategy is crucial because it is a combination of two new drugs still acts as a stumbling block. Baek So-young, Managing Director of Medical Affairs at Astellas, stated, "One might think that reimbursement would be faster for second-line or later treatments, but in reality, it takes a long time to get cancer drug reimbursement in Korea." She explained, "We applied for first-line reimbursement because we believe it provides an opportunity to save more patients." Baek added, "In terms of opening a new path for combination therapy of new drugs, the company is open to all options," and concluded, "If there is a will to reimburse Padcev, a methodological agreement can be reached through negotiation. As of now, the company has not set any restrictions."
- Policy
- MOHW proposes an alternative to law on generic substitution
- by Lee, Jeong-Hwan Aug 19, 2025 06:11am
- A revision to the Pharmaceutical Affairs Act that would allow pharmacies to report generic substitution to the Health Insurance Review and Assessment Service (HIRA) through its work portal is likely to pass the National Assembly. The background to the sudden increase in the likelihood of legislation is that the Ministry of Health and Welfare submitted its own alternative proposal to the National Assembly regarding the bill to simplify generic substitution by pharmacies and changed its position to conditional approval (acceptance) on the premise that its alternative proposal would be adopted. If the National Assembly accepts the alternative proposal submitted by the MOHW, the simplification of post-notification of generic substitution will be legislated in the Pharmaceutical Affairs Act, a law, rather than as an Enforcement Rule, a subordinate law. The alternative submitted by the MOHW stipulates that the Minister of Health and Welfare shall be able to support related administrative tasks, such as post-notification of generic substitution, and that such support tasks may be entrusted to the Health Insurance Review and Assessment Service. Rather than specifying the method of post-notification of generic substitution in the law, the proposal broadly stipulates that the MOHW shall provide support and legalize the framework for entrusting the MOHW's duties to HIRA. On the other hand, HIRA maintained its cautious stance, citing concerns for patient safety in the event of an accident involving a medication misadventure and the lack of legal grounds for entrusting the task to HIRA. This follows the Ministry of Health and Welfare’s submission, on the 18th, of its opinion on the amendment to the Pharmaceutical Affairs Act (aimed at simplifying generic substitution) to the National Assembly Health and Welfare Committee’s First Legislative Subcommittee. The law to simplify generic substitution was proposed by Representative Young-seok Seo of the Democratic Party of Korea and Representatives Sujin Lee and Byung-deok Min of the same party. The Health and Welfare Committee plans to hold the 1st Subcommittee meeting on the 19th to review the bill. The main contents are a provision to change the term “substitute dispensing” to “generic name substitutions” and a provision to expand the scope of post-notification of substitute prescriptions to HIRA. The change in terminology provision has been strongly opposed by the medical community, including the MOHW, and it is unlikely to pass the National Assembly. The provision to simplify generic substitution by expanding the scope of post-notification was once rejected by the subcommittee after the MOHW expressed its need for cautious review. However, the MOHW submitted a legal alternative it had drafted itself to the subcommittee and changed its position to support the legislation on the condition that it be passed by the National Assembly as is. The simplification of post-notification of generic substitution is expected to take effect on February 2 next year, as MOHW has already confirmed the revision of the Enforcement Rules of the Pharmaceutical Affairs Act, and the possibility of the National Assembly revising the Pharmaceutical Affairs Act has also increased. The MOHW has once again expressed its cautious stance on legislation that would require HIRA to notify prescribing doctors of the results of post-notification of generic substitution by pharmacies. The MOHW argued that this is not in line with the purpose and scope of HIRA's establishment and that the increase in the notification period could raise concerns about the safety of drug use. The MOHW also pointed out that the amendment to the Enforcement Rules of the Pharmaceutical Affairs Act allows post-notification via HIRA's information system in addition to telephone and fax. However, in consideration of the need to clarify the legal basis for the HIRA information system currently being developed by the MOHW and the necessity of substitute dispensing in response to drug supply uncertainties, the MOHW stated that an alternative plan would be necessary to establish a basis for HIRA's policy support. So the alternative submitted by the MOHW to the National Assembly stipulates the establishment of “support for generic substitution under Article 27-2 of the Pharmaceutical Affairs Act” and, in Paragraph 1, stipulates that the Minister of Health and Welfare may provide support for matters specified by MOHW ordinances, such as support for post-notification of generic substitutions. Paragraph 2 allows the Minister of Health and Welfare to entrust HIRA with the task of providing support for post-notification of generic substitution, and paragraph 3 stipulates that the details and methods of support and other necessary matters shall be determined by MOHW ordinances.. However, HIRA maintained its cautious stance on the bill. The logic behind its need for cautious review is that if post-notification generic substitution is carried out through HIRA, the notification period will increase, and if an accident occurs due to a lack of awareness on the part of doctors, this will raise concerns about patient safety. In addition, HIRA argued that it can only perform tasks entrusted to it by other laws and regulations in accordance with Article 63 of the National Health Insurance Act, and therefore, a separate basis for entrusting tasks related to generic substitution notifications is necessary. Furthermore, it also argued that unique identification numbers in accordance with the Personal Information Protection Act, regulations on the handling of sensitive information, and provisions exempting HIRA from liability for medication misadventure are necessary. d The Korean Pharmaceutical Association in favor, Korean Medical Association and Korean Hospital Association oppose The Korean Pharmaceutical Association supported the bill. On the other hand, the Korean Medical Association and the Korean Hospital Association maintained their opposition. The Korean Pharmaceutical Association predicted that allowing HIRA to notify doctors and dentists electronically would eliminate administrative inconveniences and streamline procedures, thereby promoting generic dispensing, considering the difficulty of notifying doctors and dentists when pharmacists substitute drugs due to factors such as failure to include fax numbers or failed phone connections. The Korean Medical Association argued that pharmacists could arbitrarily change medications without the consent of doctors, which could undermine the right of doctors to prescribe medications, make it difficult to respond quickly to adverse drug reactions, and raise questions about the accountability of the dispensed generic substitutions and their side effects. The Korean Hospital Association also opposed the bill, arguing that notifying just the HIRA of generic substitutions would make it difficult for doctors and pharmacists to share information about generic substitutions, which could delay the necessary treatment for patients in cases of inappropriate generic substitutions.
- Opinion
- [Reporter's View] Clarify support for unstable supply drugs
- by Lee, Hye-Kyung Aug 19, 2025 06:11am
- The Ministry of Food and Drug Safety is reportedly preparing support measures to stabilize the supply of national essential medicines and other items with unstable supply. The MFDS initially announced its intention to specify the urgent measures required by the industry to stabilize supply, such as emergency imports, custom manufacturing, and administrative support, within the first half of this year. However, it was not specified within the first half of the year, and it is said that the authorities are still considering how to specify and disclose the measures. Support for stabilizing the supply of drugs is provided by the Pharmacuetical Management Support Team of the MFDS. The team was newly established in March 2024 after the tasks previously handled by the Pharmaceutical Policy Division within the Pharmaceutical Safety Bureau were transferred due to the growing need to respond to public health crises caused by COVID-19. The Pharmaceutical Management Support Team will operate on a temporary basis until January 31, 2027, and is currently responsible for securing a stable supply system for essential medicines, ensuring the swift and stable supply of medical products in crisis situations, as well as other related tasks. In particular, it is responsible for tasks such as the designation of essential medicines, reporting on supply disruptions or shortages, monitoring supply and demand, providing administrative support and communicating with companies, and taking measures to maintain the supply of discontinued items. In this process, the pharmaceutical industry has consistently requested that administrative support measures be clearly stipulated in writing. Support measures are necessary to ensure the continued supply of essential medicines in cases of supply disruptions, as there have been instances where price increases were implemented for certain items. Therefore, there is a growing demand for the sharing of individual cases to clarify what support can be provided for which specific items. The MFDS is also preparing guidelines that include various administrative support measures, but the problem is that it is struggling with whether to use these guidelines internally or disclose them externally. If the guidelines are disclosed externally, administrative support may be applied to all items, which would inevitably increase the administrative burden. In the case of stabilizing the supply of pharmaceuticals, support measures may vary by product, and if the guidelines are made public, some companies may raise issues regarding the varying standards. However, formalizing administrative support could be one of the most effective measures to help companies stabilize supply. Companies may need to bear the burden of administrative costs and continue producing items that could otherwise be discontinued, so it is crucial for the government to clarify the specific support it can provide. Therefore, if guidelines are established, they should be publicly disclosed rather than kept internal, and administrative support measures should be properly established through a case-by-case approach.
- Company
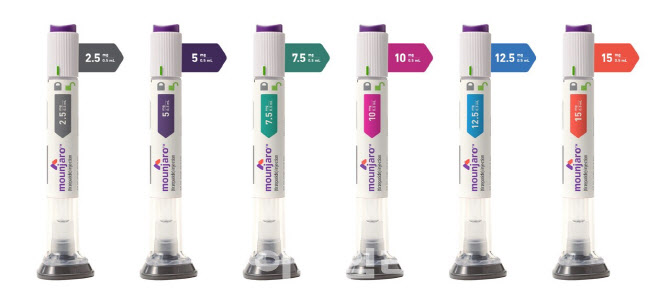
- Will Wegovy patients switch to Mounjaro? Doctors skeptical
- by Moon, sung-ho Aug 19, 2025 06:11am
- With the anticipated launch of Lilly Korea's diabetes and obesity treatment Mounjaro (tirzepatide) in mid-August, its full-scale competition with Novo Nordisk's Wegovy (semaglutide) is expected to unfold. Amid the imminent competition, the issue of whether to switch between the two drugs has emerged as a key point of contention in clinical settings. #According to industry sources on the 11th, Lilly Korea plans to launch Mounjaro Pre-filled Pen (tirzepatide, hereinafter referred to as Mounjaro) 2.5 and 5 mg for patients with type 2 diabetes and obesity in Korea in mid-August. Mounjaro is currently used in Korea as an adjunct to diet and exercise (as monotherapy or combination therapy) to improve blood glucose control in adult patients with type 2 diabetes, as well as in adult patients with obesity (initial BMI ≥ 30 kg/m2) or one weight-related comorbidity (hypertension, dyslipidemia, type 2 diabetes, obstructive sleep apnea, or cardiovascular disease) for chronic weight management as an adjunct to a low-calorie diet and exercise therapy. For both indications, the recommended starting dose is 2.5 mg once weekly (for treatment initiation, not for blood glucose control or weight management), followed by 5 mg once weekly starting at week 4. If additional dose adjustment is required, increase by 2.5 mg after at least 4 weeks of treatment at the current dose, with a maximum dose of 15 mg once weekly. The attention is now focused on the competition between Wegovy and Mounjaro in clinical practice. A key point of interest in this process is whether patients previously treated with Wegovy will switch to Mounjaro. Although there are no direct comparative studies, based solely on the results revealed during the development process, the weight loss effect of Mounjaro appears to be more pronounced. Considering this, some patients who were previously receiving Wegovy may express a willingness to switch to Mounjaro. In response, Lilly Korea stated that the decision to change the type of medication or treatment method prescribed to patients should be made through discussion between healthcare professionals and patients. A representative from Lilly Korea stated, “We have not conducted any studies on switching from GLP-1 RA to Mounjaro for chronic weight management on our own. However, we have a Phase IV clinical trial called SURPASS-SWITCH-2, which involved switching from GLP-1 RA (semaglutide, dulaglutide, or liraglutide) to Mounjaro in adult patients with type 2 diabetes for 12 weeks. The study was not designed to provide long-term results, efficacy comparisons, or optimal switching strategies, as these may vary depending on individual characteristics." In clinical practice, not many patients are expected to switch from Wegovy to Mounjaro, considering how the initial dose of Mounjaro is 2.5 mg. Chul-Jin Lee, President of The Korean Society For the Study of Obesity (Joeun Family Clinic), said, “explained, “Just because Mounjaro has been launched does not mean that patients who were previously receiving Wegovy will easily switch treatments in the short term. This is because patients who were receiving high doses of Wegovy may experience relatively lower efficacy when switching to 2.5 mg of Mounjaro.” Lee said, “If Mounjaro is released, it should be offered first to new patients. The 2.5 mg dose has an advantage in terms of price for patients as it is not covered by insurance, but for the 5 mg dose and above, the volatility of the range of the non-reimbursed price must also be considered.” Meanwhile, the supply price for Mounjaro has recently been finalized. The currently known domestic supply prices for Mounjaro are KRW 278,066 for 2.5mg, KRW 369,307 for 5mg, and KRW 521,377 for both 7.5mg and 10mg. The dosage forms to be launched in Korea this month are 2.5mg and 5mg. For reference, when Novo Nordisk launched Wegovy in Korea last year, the supply price was set at KRW 372,025 per month regardless of dosage. Accordingly, patients who receive Wegovy as a non-reimbursable drug, mainly at outpatient clinics, must pay an average of KRW 500,000 to 600,000 per month. Considering this, when the supply price is taken into account, the non-reimbursed price of 2.5 mg of Mounjaro is expected to be cheaper than Wegovy, but 5 mg is expected to be priced similar to the monthly cost of Wegovy.
- Company
- PARP inhibitor 'Lynparza' reattempts prostate cancer reimb
- by Eo, Yun-Ho Aug 18, 2025 06:03am
- Product photo of LynparzaAttention is focused on whether there will be progress in the expanded reimbursement of the PARP inhibitor 'Lynparza' for prostate cancer. According to industry sources, AstraZeneca Korea's PARP (Poly ADP-ribose polymerase) inhibitor Lynparza (olaparib) is expected to be considered for the Health Insurance Review & Assessment Service (HIRA)'s Cancer Disease Review Committee next month (September). The indications for review are two: ▲Treatment of adult patients with BRCA-mutated metastatic castration-resistant prostate cancer (mCRPC) who have experienced disease progression after treatment with a novel hormonal agent ▲Combination therapy with abiraterone and prednisolone in adult patients with newly diagnosed mCRPC who have not received prior chemotherapy. This is Lynparza's second attempt to get reimbursement for its prostate cancer indication. AstraZeneca had previously tried in 2022 to get reimbursement for BRCA-mutated prostate cancer, but the application was withdrawn after failing to reach an agreement during the post-Cancer Disease Review Committee procedure. Therefore, it remains to be seen if this second round of reimbursement discussions will be successful. Meanwhile, the efficacy of Lynparza in prostate cancer has been proven through two Phase 3 trials: the PROfound study and the PROpel study. A subgroup analysis of the PROfound study in mCRPC patients with BRCA1/2 mutations showed that in patients who had progressed after prior abiraterone or Xtandi (enzalutamide) treatment, Lynparza significantly improved radiographic progression-free survival (rPFS) to a median of 9.8 months, compared to 3.0 months for the control group. Overall survival (OS) was also improved, with a median of 20.1 months versus 14.4 months for the control group. The PROpel study evaluated the combination therapy of Lynparza and abiraterone in mCRPC patients who had not received prior chemotherapy. The study showed that the combination therapy reduced disease progression or the risk of death by 34% compared to abiraterone monotherapy. The median rPFS was extended by 8.2 months in the combination group, reaching 24.8 months compared to 16.6 months in the abiraterone monotherapy group.
- Company
- Four CAR-T therapies to compete for 'Kymriah's mkt position'
- by Whang, byung-woo Aug 18, 2025 06:02am
- With the third new CAR-T therapy, 'Yescarta,' obtaining marketing authorization in Korea, market competition for treating relapsed or refractory Diffuse Large B-cell Lymphoma (DLBCL) is anticipated. With Kymriah (tisagenlecleucel) from Novartis Korea being the only option currently reimbursed, the impact of Yescarta, which has shown prominence in the global CAR-T market, is highly anticipated. Furthermore, with the expected approval of Curocell's next-generation CAR-T therapy, Rimqarto, this year, various factors are expected to be considered in the choice of DLBCL treatments. Product photo of YescartaThe Ministry of Food and Drug Safety (MFDS) announced the marketing authorization for Gilead Sciences Korea's Yescarta (axicabtagene ciloleucel) on the 13th. Yescarta received authorization for the treatment of 'adult patients with Diffuse Large B-cell Lymphoma (DLBCL) who relapse or are refractory within 12 months of first-line chemoimmunotherapy' and 'adult patients with relapsed or refractory DLBCL and Primary Mediastinal B-cell Lymphoma (PMBCL) after two or more lines of systemic therapy.' Administration is required to be performed only at certified medical institutions, and certified medical institutions must have tocilizumab readily available on-site. The authorization also specifies that at least two doses of tocilizumab per patient must be secured before Yescarta administration in case treatment for Cytokine Release Syndrome (CRS) is needed. Yescarta is currently a leading therapy in the CAR-T treatment market. This is because Yescarta has secured a broader range of indications than Kymriah, which is limited to third-line therapy. First approved by the U.S. FDA as a third-line therapy in October 2017 and by the EU in 2018, Yescarta has since expanded its scope to include second-line therapy. In 2021, it became available to treat follicular lymphoma. In terms of sales, its revenue surged by 67%, from $695 million (approximately KRW 966.4 billion) in 2021 to $1.16 billion (approximately KRW 1.6127 trillion) in 2022. The growth continued, reaching $1.498 billion (approximately KRW 2.0827 trillion) in 2023, a 29% increase from the previous year. However, since the rapid growth of late entrants like Carvykti (ciltacabtagene autoleucel, Janssen) and Breyanzi (lisocabtagene maraleucel, BMS) starting last year, the pace of revenue increase has slowed. In its recently announced Q2 sales for this year, Gilead reported that Yescarta recorded $393 million (approximately KRW 550 billion), a 5% decrease from the previous year. The reason for the anticipation surrounding Yescarta is its indication. Kymriah's indication is for 'adult patients with relapsed or refractory DLBCL after two or more lines of systemic therapy.' For this reason, Gilead Sciences Korea is reportedly planning to prepare for reimbursement for the newly approved second-line therapy, rather than the third-line therapy, as Kymriah is already reimbursed for the third-line setting, making reimbursement for Yescarta at this stage less difficult. While new drugs of bispecific antibodies are receiving approval and reimbursement, CAR-T therapies still hold a preemptive treatment status. If Yescarta receives reimbursement, it is expected to quickly establish itself in the domestic market, given its global influence. Curocell Prepares for Rapid Commercialization…"Efficacy and Supply Still the Advantages" Another variable is that Curocell's Rimqarto is awaiting approval. Rimqarto has been selected as the second drug for the Ministry of Health and Welfare's 'Concurrent Application-Evaluation-Negotiation Pilot Program' and is currently undergoing review by the MFDS. While there are observations that the approval might be delayed beyond the initially expected third quarter due to delays in the review process, as it is the first domestically developed next-generation CAR-T therapy in Korea, the prevailing view is that approval will likely happen within the year. Once approved, it is expected to have an advantage in reimbursement entry because the reimbursement evaluation and price negotiation will be conducted simultaneously with the MFDS approval application process. In particular, the company is preparing for rapid commercialization by establishing a comprehensive solution, 'CUROLINK,' which tracks and manages the entire process from prescription to administration in real time. However, Rimqarto must keep Yescarta's approval in check, as it has the same indication as Kymriah. Concerning this, Curocell's position is that its competitiveness will remain valid because, when compared to Curocell's Rimqarto, Yescarta, and Kymriah are still inferior in terms of supply period, efficacy, and safety in the domestic market. A Curocell official stated, "Yescarta's entry into the Korean market and the emergence of other competing products like bispecific antibodies are always open possibilities," and added, "Yescarta still requires sending the patient's blood overseas for CAR-T manufacturing and then re-importing it into Korea, placing it in an inferior position compared to Curocell's Rimqarto in terms of rapid supply." Furthermore, according to an IR document released by Curocell, Yescarta showed a complete remission in 54% of patients who participated in the clinical trial. In contrast, Rimqarto's clinical trial showed a complete remission in 67% of participants. The Curocell official explained, "Yescarta's CR rate of 54% was far superior to Kymriah's 40%, which led to a decrease in Kymriah's sales and an increase in Yescarta's sales. However, when Breyanzi, with a CR rate of 53%, entered the market with a similar CR rate to Yescarta, it began to share the market, causing a slight decrease in Yescarta's sales." And added, "Considering this, Rimqarto's CR rate of 67% is higher than both Yescarta and Breyanzi. Given that this is a one-time reimbursed treatment for late-stage cancer patients, We believe Rimqarto will have a competitive advantage in the market."
- Company
- New Praluent dosage form available at general hospitals
- by Eo, Yun-Ho Aug 18, 2025 06:02am
- A new dosage form of the PCSK9 inhibitor ‘Praluent’ may be prescribed at general hospitals in Korea. According to industry sources, a pen formulation of Sanofi-Aventis Korea's primary hypercholesterolemia and mixed dyslipidaemia treatment Praluent (alirocumab), Praluent Pen 300mg inj, has been approved by the Drug Committees (DCs) of tertiary hospitals in Korea, including Seoul National University Hospital, Seoul Asan Hospital, and Sinchon Severance Hospital. Praluent Pen Inj 300mg maintains similar LDL-C lowering effects to the existing dosages while enabling administration at four-week (Q4W) intervals, thereby improving treatment convenience and patient compliance. This drug is the only PCSK9 inhibitor in Korea available in three doses (75 mg, 150 mg, and 300 mg), and all doses are indicated for reducing cardiovascular risk in patients with atherosclerotic cardiovascular disease (ASCVD). PK/PD studies confirmed the LDL-C-lowering efficacy of Praluent Pen 300mg, with LDL-C reduction effects observed as early as day 3 after a single dose of Praluent Pen 300mg injection, reaching a maximum average reduction of 73.7% by day 22. This LDL-C-lowering effect was maintained for up to 43 days, demonstrating a longer duration of effect compared to the existing Praluent 75mg Pen (8 days) and 150mg (15 days). Praluent 300 mg has been listed for reimbursement in Korea since April. As a result, Praluent Pen Inj may be used with reimbursement when administered in addition in ▲patients with primary hypercholesterolemia and mixed dyslipidemia who have received insufficient response to combination therapy with statins and ezetimibe (LDL-C levels not reduced by at least 50% from baseline or LDL-C ≥ 100 mg/dL) and statin intolerance ▲Patients with atherosclerotic cardiovascular disease who have received maximum tolerated doses of statins and ezetimibe but have had an inadequate response (LDL-C levels not reduced by at least 50% from baseline or LDL-C ≥ 70 mg/dL). In a Phase III trial (CHOICE 1) involving patients who were taking the maximum tolerated dose of statins, patients who were taking the maximum tolerated dose of statins in combination with other lipid-lowering agents, and patients who were not taking statins, the starting does of Praluent Pen 300 mg Inj demonstrated efficacy, long-term safety profile, and tolerability. According to the study, at week 24, the LDL-C reduction effect of Praluent 300 mg Inj plus statin therapy compared to placebo was 58.8%, showing a statistically significant result compared to the placebo group (0.1% reduction). In the case of its indication for atherosclerotic cardiovascular disease, the drug demonstrated efficacy iin the ODYSSEY OUTCOMES study. The study included 18,924 adult patients with acute coronary syndrome (ACS), including myocardial infarction and unstable angina. The results showed that Praluent significantly reduced the risk of major adverse cardiovascular events (MACE) by 15% compared to the placebo group. A trend toward a reduction in all-cause mortality was also observed as a secondary endpoint, which was nominally significant in hierarchical statistical testing. In addition, y, LDL-C achieved its maximum reduction effect at week 4 of Praluent treatment and maintained an average reduction of 54.7% after 4 years of treatment. LDL-C reduction was observed in 89% of patients receiving high-intensity statins in combination with Praluent.