- LOGIN
- MemberShip
- 2026-04-03 12:18:25
- Company
- Metabolic disease drug development landscape evolves
- by Son, Hyung Min Jul 01, 2025 06:02am
- Innovative efforts in the development of new drugs for metabolic disorders such as obesity and diabetes are steadily gaining traction in Korea and abroad. The success of standalone GLP-1 therapies has driven the evolution toward multi-hormonal agents, and recently, major global pharmaceutical companies and biotech firms are accelerating new drug development by targeting not only GLP-1, GIP, and glucagon combinations but also new metabolic hormones such as FGF21 and IGF-1. In essence, a multi-target approach that goes beyond simple weight loss to regulate energy metabolism, insulin sensitivity, and liver fat improvement is becoming a reality. Development of multi-agonists starts with Mounjaro... Biomed announces Phase II trial According to industry sources on the 30th, the US pharmaceutical and biotech company Biomed Industries recently announced the results of its Phase II clinical trial for NA-931, an oral quadruple receptor agonist candidate for obesity and metabolic disorders. The results were presented at the American Diabetes Association's annual diabetic conference (ADA 2025) that was held in Chicago from the 21st to the 24th. NA-931 is a small-molecule drug that acts on four receptors: glucagon-like peptide (GLP-1), gastric inhibitory peptide (GIP), glucagon (GCG), and insulin-like growth hormone type 1 (IGF-1). Recently, research has also been conducted on quadruple receptor agonists that utilize additional metabolic-related hormones such as IGF-1, FGF21, and PYY, in addition to GLP-1, GIP, and GCG. FGF21 is a hormone produced in the liver that is involved in fat oxidation, insulin sensitivity, and temperature regulation. It is gaining attention as a next-generation target due to its ability to induce metabolic improvements similar to those observed with intermittent fasting. PYY is an appetite-suppressing hormone derived from the gut that can contribute to enhancing satiety by interacting with GLP-1. The Phase II clinical trial presented at the conference was a 13-week, randomized, double-blind, placebo-controlled, parallel-group study involving 125 adults with obesity (BMI ≥ 30 kg/m²) or overweight (BMI ≥ 27 kg/m²). The primary endpoints were the safety, tolerability, and weight loss efficacy of the investigational drug NA-931. Results showed that the group administered NA-931 at a dose of 150 mg once daily experienced an average weight reduction of 13.8% compared to baseline, with a statistically significant reduction of 12.4 percentage points compared to the placebo group. The incidence of treatment-emergent adverse events was also favorable. Gastrointestinal (GI) adverse events were mostly mild, with nausea and vomiting observed in 7.3% of participants and diarrhea in 6.3%. No muscle mass reduction was reported, and there was no clinically significant difference in the incidence of GI-related adverse events among subjects treated with NA-931 compared with placebo. The researchers explained, “NA-931 appears to induce weight loss while preserving muscle mass and has a lower incidence of side effects than existing drugs. It can be regarded as a viable alternative that can improve the shortcomings of existing GLP-1 class treatments.” Development of quadruple receptor agonists continue… to treat MASH in addition to obesity Clinical studies of quadruple receptor agonists targeting indications other than obesity are also actively underway. Among domestic companies, Wonjin Biotechnology has entered the clinical trial phase for a quadruple receptor agonist. Wonjin Biotechnology received approval in the US last year for the Investigational New Drug (IND) application for its candidate compound ‘OGB21502’ for the treatment of metabolic dysfunction-associated steatohepatitis (MASH) and fibrosis. OGB21502 is an innovative drug candidate that combines GLP-1, glucagon, FGF21, and an IL-1 receptor agonist (IL-1RA), an immune modulator, to simultaneously regulate metabolic disorders and chronic inflammation. OGB21502 induces appetite suppression and energy metabolism activation through GLP-1 and glucagon receptors while promoting lipid metabolism via FGF21 action. Additionally, by blocking IL-1 signaling involved in inflammation, the company expects it to demonstrate a multi-layered therapeutic effect by preventing inflammatory exacerbation of metabolic diseases. According to Wonjin Biotechnology, there is growing evidence that therapies blocking IL-1 signaling are effective in alleviating liver fibrosis. In fact, anakinra, a recombinant IL-1 receptor antagonist, has been shown in multiple studies to inhibit NLRP3 activation and reduce hepatic stellate cell (HSC) activity and blood-based fibrosis markers. In preclinical studies, OGB21502 demonstrated a reduction in lipid markers, including cholesterol and triglycerides, as well as the expression of fibrosis-related markers, compared to semaglutide and FGF21 analogs in obese mouse models. In a mouse model of non-alcoholic fatty liver disease (NAFLD), liver tissue staining analysis revealed that OGB21502 significantly reduced the non-alcoholic fatty liver disease activity score (NAS) and improved steatosis and lobular inflammation.
- Company
- AstraZeneca Korea launches ‘Lung Health Checkbus’ campaign
- by Whang, byung-woo Jul 01, 2025 06:00am
- Launch ceremony for AstraZeneca Korea AstraZeneca Korea announced on the 30th that it held a ceremony for the launch of its ‘Lung Health Checkbus’ campaign at the COEX Square in Seoul on the 27th. The campaign aims to help people detect lung nodules that they are not aware of at an early stage by operating buses equipped with AI-based chest X-ray imaging nationwide. AstraZeneca Korea, which has been making various efforts to create a world where lung cancer is no longer a cause of death, has partnered with the Korea National Tuberculosis Association and medical AI solution company Maihub to operate the ‘Lung Health Checkbus’ nationwide. Lung cancer is the leading cause of cancer-related deaths in Korea, according to 2023 data. Over the 5-year period from 2018 to 2022, the relative survival rate was 79.8% when the cancer was detected at an early stage, but the rate dropped sharply to 12.9% when the cancer had spread to distant parts of the body. However, it has been reported that more than 40% of patients are diagnosed with cancer at an advanced stage of metastasis, emphasizing the importance of regular screening. Low-dose chest CT is an effective screening method that can accurately detect lung cancer and reduce mortality, and AI-equipped chest X-rays are more effective than conventional X-rays in detecting lung nodules. According to a study comparing the lung nodule detection rates of AI-equipped chest X-rays and conventional X-rays at a single institution in Korea, the lung nodule detection rate in the AI group was more than twice that of the non-AI group. Se-Hwan Chon, General Manager of AstraZeneca Korea, said, “Lung cancer can affect anyone, so lung cancer screening is necessary even for non-smokers. In particular, lung cancer has a significantly higher survival rate when detected early, so it is important to detect it early through regular screening.” On the same day, AstraZeneca Korea signed a three-party memorandum of understanding (MOU) with the Korea National Tuberculosis Association and Maihub to ensure the successful operation of this campaign. Many citizens had chest X-rays taken at the lung health check bus set up on-site and received reports analyzed by artificial intelligence (AI) to check their lung health status for themselves. Min-Seok Shin, Chair of KNTA, said, “We find it meaningful that KNTA can expand its social responsibility beyond respiratory diseases to a broader range of diseases through the campaign. We plan to continue supporting customized health management programs to ensure that everyone, including medically vulnerable groups, can easily check their lung health. Hyuck Yang, CEO of Maihub, said, “This campaign is a meaningful example of public and private sectors coming together to create an AI-based chest X-ray examination environment that is easily accessible to everyone, going beyond simply providing technology. Maihub will contribute to the early detection of lung nodules through AI-based image interpretation solutions and strive to build a digital healthcare ecosystem.” Meanwhile, AstraZeneca Korea is a member of the Lung Ambition Alliance (LAA), a global non-profit collaboration organization, and is conducting various lung cancer awareness improvement activities to create a future where lung cancer is no longer a cause of death.
- Company
- New K-drugs for metabolic diseases make international debut
- by Son, Hyung Min Jun 30, 2025 06:07am
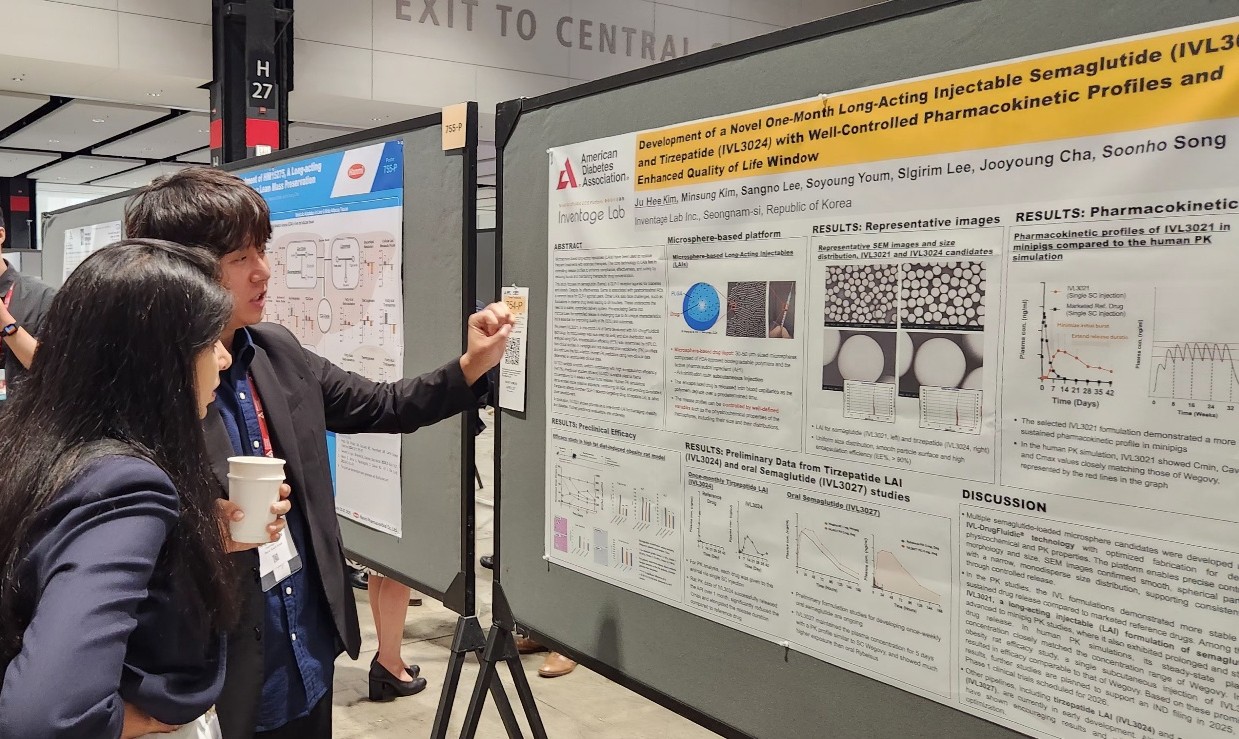
- Major Korean pharmaceutical and biotechnology companies have signaled their full-scale entry into global clinical trials, presenting new drug development results at overseas conferences. The companies presented their achievements in developing new drugs for various metabolic diseases, including obesity, type 2 diabetes, and metabolic dysfunction-associated steatohepatitis (MASH). Although most of the data disclosed is focused on early clinical or preclinical trials, the companies are attempting to diversify their mechanisms of action with triple agonists and oral small molecule formulations. Hanmi reveals triple agonist data... Oral and long-acting injectable formulations are also under development 30According to industry sources, the American Diabetes Association Diabetes Conference was held in Chicago, USA, from the 21st to the 24th. At the conference, various domestic companies, including Hanmi Pharmaceutical, Ildong Pharmaceutical, Dong-A ST, and Inventage Lab, revealed the clinical results of their novel drug candidates. Obesity drugs are rapidly emerging as a global R&D trend. With Novo Nordisk and Lilly's GLP-1-based obesity treatments becoming global blockbuster drugs, latecomers are also intent on developing their versions. Major domestic companies are conducting clinical studies on GLP-1 agonists in various areas, including obesity and MASH with drugs that have different methods of administration or focus on the quality of weight loss effects. Hanmi Pharmaceutical announced the results of its Phase I clinical trials and preclinical data for HM15275, a GLP-1 class triple agonist, and HM17321, a UCN2-based candidate substance, at the conference. First, Hanmi Pharmaceutical announced the results of the Phase I clinical trial for HM15275. HM15275 is a new obesity drug candidate that acts on glucagon-like peptide (GLP-1), gastric inhibitory polypeptide (GIP), and glucagon (GCG). No new drug with this mechanism targeting all three has been commercialized to date. The Phase I clinical trial was conducted on 74 healthy and obese adults. HM15275 was administered subcutaneously once a week for four weeks, followed by an evaluation of the candidate drug’s safety, tolerability, pharmacokinetics, and pharmacodynamics. The results confirmed HM15275’s tolerability and safety. Specifically, the average weight loss rate at day 29 in the highest dose group was 4.8%. At day 43, the maximum weight loss rate was 10.6%. Preclinical data for HM15275 demonstrated greater weight loss efficacy compared to semaglutide (brand name Wegovy) and tirzepatide (Zepbound), which are currently marketed as obesity treatments. In animal models, switching from tirzepatide to HM15275 resulted in additional weight loss effects. Hanmi Pharmaceutical also announced the results of preclinical studies on HM17321, a new drug candidate that simultaneously targets weight loss and muscle gain. HM17321 is a UCN-2 analogue that selectively targets the CRF2 (corticotropin-releasing factor 2) receptor rather than GLP-1 or other incretin receptors. It is being developed as an innovative first-in-class drug that selectively reduces fat while increasing muscle mass. HM17321 demonstrated weight loss effects and improved body composition in both mouse models and non-human primate models. Yunovia, a subsidiary of Ildong Pharmaceutical Group specializing in new drug research and development, is conducting Phase I clinical trials for ID110521156, a GLP-1 receptor agonist class new drug candidate targeting metabolic disorders such as diabetes and obesity. ID110521156 is a low-molecular-weight compound-based drug, and the company aims to develop it as an oral synthetic new drug for diabetes and obesity with distinct advantages such as superior productivity and excellent ease of use over existing representative treatments like peptide injections. Previously, Yunovia confirmed the efficacy of insulin secretion and blood glucose control through preclinical efficacy and toxicity evaluations. It also demonstrated superior safety compared to competing drugs in the same class and confirmed the drug's characteristics in a recently completed Phase 1 single-ascending dose (SAD) trial. According to the study poster presented at the conference, in the single-dose escalation trial, ID110521156 demonstrated good tolerability with fewer gastrointestinal side effects across the entire effective dose range, unlike existing GLP-1 class drugs. Inventage Lab also introduced preclinical data for its 1-month long-acting injectable formulations ‘IVL3021’ and ‘IVL3024’ based on semaglutide and tirzepatide, as well as its oral semaglutide formulation ‘IVL3027.’ After GLP-1 class obesity treatments such as Saxenda, Wegovy, and Zepbound emerged as global blockbuster drugs, the pharmaceutical industry has been actively pursuing formulation changes. The existing drug Saxenda requires a once-daily injection, while Wegovy and Zepbound require weekly injections. Oral formulations or long-acting injectables are expected to gain a competitive edge in terms of convenience of administration if commercialized. According to the company's preliminary preclinical results, IVL3021 showed stable drug release in the blood over a one-month period. Also, the long-acting injectable suppressed initial over-release and maintained stable drug release. IVL3027 demonstrated high bioavailability compared to existing oral formulations and sustained drug release over a one-week period. Poster presentation by Inventage Lab (Source=Inventage Lab). MASH clinical trial results also announced Dong-A ST and its subsidiary MetaVia announced the results of non-clinical studies on DA-1241, which is being developed as a treatment for MASH, and combination therapy that uses efruxifermin, a fibroblast growth factor (FGF21) analog. Metabolic dysfunction-related fatty liver disease was previously referred to as non-alcoholic steatohepatitis (NASH), but overseas academic societies such as the American Association for the Study of Liver Diseases have decided to change the name to metabolic dysfunction-associated steatohepatitis (MASH). To date, Madrigal's rezdiffra is the only new drug for MASH that has cleared regulatory hurdles. The U.S. Food and Drug Administration (FDA) approved rezdiffra in March last year for the treatment of adult patients with non-cirrhotic MASH in combination with diet and exercise. Rezdiffra is a selective thyroid hormone receptor (THR)-β agonist designed to target the core pathophysiological mechanisms of MASH within the liver. The pharmaceutical industry is also developing new drugs for MASH that target not only THR-β but also GLP-1 and FGF21, which influence lipid metabolism. DA-1241 is a synthetic new drug that activates GPR119. Preclinical results have confirmed that DA-1241 improves blood sugar and lipid levels and directly acts on the liver to improve inflammation and fibrosis, making it a promising candidate for MASH treatment. A Phase 2a clinical trial targeting patients with estimated MASH was completed in December last year. Efruxifermin is a recombinant protein designed based on FGF21 (Fibroblast Growth Factor 21), a hormone secreted by the liver. FGF21 is involved in energy consumption and the regulation of glucose and lipid metabolism in the body and is used as a target for the development of treatments for MASH, obesity, diabetes, and other conditions. According to the study results presented at the conference, in the DA-1241+efruxifermin combination therapy group, approximately 94% of subjects showed an improvement of 2 points or more in NAS (Non-Alcoholic Steatohepatitis Activity Score) compared to baseline. Additionally, the DA-1241+efruxifermin group showed a significant reduction in liver fibrosis area compared to the MASH control group that did not receive combination therapy, and in some individuals, a decrease in fibrosis stage was observed compared to pre-treatment levels. Dong-A ST is currently conducting clinical studies on the combination therapy of DA-1241 with semaglutide, a GLP-1 agonist, in addition to a Phase II clinical trial on DA-1241 as monotherapy.
- Company
- Adempas may be prescribed at general hospitals in KOR
- by Eo, Yun-Ho Jun 30, 2025 06:06am
- Adempas, a new treatment for pulmonary arterial hypertension that has emerged after a long wait, is now available for prescription at general hospitals in Korea. According to industry sources, Bayer Korea's Adempas (riociguat) has been approved by the Drug Committee (DC) of tertiary hospitals in Korea, including Samsung Medical Center and Seoul National University Hospital. As it has been listed for reimbursement since this month (June), the number of medical institutions that can prescribe it is expected to continue to increase. Adempas was approved in Korea as an orphan drug in June 2014 and is available in 5 dosage forms. It is indicated for: ▲Improvement of exercise capacity in adult patients with chronic thromboembolic pulmonary hypertension (CTEPH, WHO Group 4) who are unable to undergo surgery or who have persistent or recurrent symptoms after surgery ▲Improvement of exercise capacity adult patients with pulmonary arterial hypertension (WHO Group 1) who are classified as having functional class II or III. In particular, it was known as the first new drug for CTEPH. CTEPH is caused by patients who develop chronic pulmonary embolism, which leads to fibrotic stenosis and occlusion, resulting in pathological vascular remodeling and increased resistance in the pulmonary artery. CTEPH is a chronic disease that causes progressive dyspnea and right heart dysfunction, which weakens the heart. Symptoms include dyspnea, fatigue, chest pain, dizziness, peripheral edema, cough, and hemoptysis, which reduces the patient’s quality of life. Ultimately, it can progress to heart, kidney, and liver failure, which can lead to death. Meanwhile, Adempas is a stimulator of soluble guanylate cyclase (sGC), an enzyme found in the heart and lungs, and its efficacy has been confirmed in two Phase III clinical trials in patients with chronic thromboembolic pulmonary hypertension. Results showed improvement in exercise capacity, which was the primary endpoint, and good tolerability. No unexpected adverse reactions were reported. In the CHEST-1 study, when comparing the 6-minute walking test (6MWT) results after 16 weeks with the baseline, results showed that the group of patients who received riociguat showed statistically significant improvement compared to the group of patients who received placebo. In the PATENT-1 study, the change in the 6MWT score after 12 weeks of treatment, showed statistically significant improvement in the riociguat arm compared to placebo, meeting the primary endpoint.
- Opinion
- [Reporter's View] Divisional restructuring needed
- by Lee, Hye-Kyung Jun 30, 2025 06:06am
- During his candidacy, President Lee Jae-myung pledged to designate the biotech industry as a cutting-edge sector and build it as the future growth engine, aiming to position South Korea as one of the world's five strongest biotech countries. Based on an analysis that the previous government was short on investment in the pharmaceutical and biotech industry, President Lee will likely plan to implement policies geared at establishing special funds and fostering talents with expertise to strengthen national investment and responsibility in the pharmaceutical and biotech industry. Firstly, there will be expanded support at the R&D center, focusing on venture technologies. To achieve this, regulatory support is crucial. To set a goal of becoming a strong biotech country, aggressive R&D must be pursued. Additionally, supporting measures for venture firms with less regulatory experience are also necessary. Celltrion, for instance, which successfully overtook the No. 1 place in exporting biopharmaceuticals in South Korea, was established in 2002 as a biotech venture company. Therefore, when aggressive R&D takes place, there will be more venture firms with fewer years of regulatory experience that will robustly grow. The problem lies in whether the Ministry of Food and Drug Safety (MFDS), which is responsible for providing regulatory support to make South Korea one of the top five global biotech powerhouses, can properly fulfill its role. In particular, the division responsible for pharmaceutical approvals is operating as a temporary task force (TF), with a fixed staff of only seven people managing all approval-related tasks for biopharmaceuticals, Korean traditional medicines (han-yak), and quasi-drugs. Although the current government has promised active investment in biotech, it remains uncertain to determine how much will be allocated for pre-registration or applications for new drug approval. Furthermore, the government implemented an innovative measure to shorten the new drug approval duration from 420 to 295 days this year, which also includes biopharmaceuticals. According to the number of approved new drugs by year, the number of chemical medicines decreased from 29 in 2023 to 11 in 2024. In contrast, the number of biopharmaceuticals increased during the same period, from 8 to 12 cases. For biopharmaceuticals, which are often high-priced, cutting-edge therapies for rare·intractable diseases or cancers with significant medical demand, the approval process is complex. If the approval division isn't functioning properly, it becomes challenging to ensure swift internal and external communication or make highly reliable decisions based on scientific·legal reviews. Related to this, it's puzzling why the Pharmaceutical Approval Management Division remains a task force (TF). This bio approval TF was established in May 2024 when the Vice Minister's direct reporting divisions for overall approval management and advanced product approval were reorganized and redistributed across three bureaus: Pharmaceutical Safety Bureau, Biopharmaceuticals and Herbal Medicines Bureau, and Medical Device Safety Bureau, to strengthen the linkage between medical product approvals and policy. While this reorganization converted two existing divisions into three, resulting in 'Approval Divisions' for pharmaceuticals and medical devices, only bio approvals were structured as a "team"-based TF. Reportedly, MFDS faced limitations in creating new official divisions under its organizational regulations, leading to the establishment of permanent divisions for pharmaceuticals and medical devices, which handle a relatively higher volume of public complaints and regulatory support). Yet, it's difficult to comprehend. At the same time, the bio-related function was set up as a temporary task force. This is particularly difficult to understand given that becoming a biotech powerhouse has been a national priority since the previous administration, and it's a commitment significant enough to be a presidential pledge in the current government. The Pharmaceutical Approval Management Division serves as the manager for the entire approval process and acts as a communication channel between external stakeholders and the MFDS. Operating as a temporary organization with only seven full-time staff means that even the absence of a single employee can jeopardize the swift processing of applications and approvals. If the approval department doesn't function properly, it becomes difficult to ensure quick internal and external communication or make reliable decisions based on scientific and legal reviews. While the current proportion of pharmaceuticals in the overall medical product industry in South Korea may still be low, there's significant potential for further growth, depending on the role of the President Lee government. To accelerate the commercialization of domestically developed biopharmaceuticals and secure a leading global position, the MFDS will likely need to reorganize its structure to provide proactive regulatory support.
- Policy
- More generic 'Vimovo' drugs with naproxen+PPI enters the mkt
- by Lee, Tak-Sun Jun 30, 2025 06:05am
- Product photo of the original drug Generic drugs containing the same active ingredients as the 'Vimovo' (naproxen+esomeprazole magnesium trihydrate), a combination of an anti-inflammatory drug and an anti-ulcer agent, are set to be released. A generic has not been approved since Chong Kun Dang's 'Naxen S Tab' was approved in 2024. Considering the characteristics of a generic containing two types of active ingredients, proving a pharmaceutical equivalence test on each active ingredient may have been challenging. Furthermore, analysis suggests new product entry to this market poses a challenge due to 'Naxozole,' a salt-changed product that was launched at a relatively lower price, with a strong presence. According to the industry on June 27, four generic Vimovo drugs, including KyungDong Pharm's 'Nasopra Tab 500/20 mg,' will be listed with reimbursement next month. KyungDong Pharm is the organizing company. The company's Nasopra Tab 500/20 mg met two types of requirements. Thus, the price was assessed at KRW 715, the same as the original Vimovo. In contrast, the drug price of the generics meeting only one requirement was set as KRW 608. These include Genuonesciences' 'Gemovo Tab 500/20 mg,' Mother's Pharmaceutical's 'Vimo M Tab. 500/20 mg,' and Dongkook Pharmaceutical's 'Exoraxen Tab 500/20 mg.' These drugs treat symptoms of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis in patients at risk of gastric or duodenal ulcers associated with NSAIDs, such as naproxen, or in those who are not adequately treated with low-dose naproxen or other NSAIDs. Joined later by four companies, products containing the same active ingredients as Vimovo have now increase from two to six. Previously, LG Chem's 'Vimovo Tab 500/20 mg' and Chong Kun Dang's 'Naxen S Tab' were the only generics available. Furthermore, Naxen S Tab has been on the market for over 10 years since its approval in 2014. Even without re-examination periods or patent barriers, generic versions have not been made available, likely due to the significant difficulty of conducting bioequivalence tests for this combination therapies. To prove bioequivalence for generic Vimovo drugs, it's necessary to compare the human absorption rates of each component, naproxen and esomeprazole. This process is complex, and achieving favorable results is challenging. Korean pharmaceutical companies have shifted their development efforts towards modified drugs that use a different salt of esomeprazole. Currently, there are five such salt-changed products available, including Hanmi Pharm's 'Naxozole Tab. 500/20 mg. Naxozole recorded KRW 25.8 billion in outpatient prescription sales last year, according to UBIST data, surpassing the original Vimovo (KRW 21.7 billion). The issue, however, is that Naxozole is relatively low-priced, at KRW 445 per tablet, which is the cheapest among competing products. Naxen S, a generic version of Vimovo, is relatively inexpensive at KRW 490 and recorded KRW 4.2 billion in outpatient prescription sales last year. In contrast, these newly reimbursed generic drugs are priced KRW 100-200 higher than Naxen S, based on the estimation criteria. Therefore, it's uncertain whether they will exhibit drug price competitiveness against Naxozole and Naxen S in the market. However, an analysis suggests that if they enter the market with high CSO (Contract Sales Organization) fees based on their higher drug prices, they could increase their market share. Amid other generic companies also preparing for market entry, attention is now focused on the market performance of the products from KyungDong Pharm and the other three companies that have recently obtained reimbursement.
- Policy
- Polivy granted partial reimbursement after 5 years
- by Lee, Tak-Sun Jun 30, 2025 06:05am
- Roche’s Polivy (polatuzumab vedotin), a treatment for diffuse large B-cell lymphoma (DLBCL) that is currently non-reimbursed in Korea, has been added to the reimbursement list as a part of combination therapy. With the listing, the other drugs used in the combination, excluding Polivy, will be reimbursed. This measure is in accordance with the partial reimbursement policy for combination cancer therapies that was implemented in May. On the 26th, the Health Insurance Review and Assessment Service announced the reimbursement criteria for anticancer drugs and announced 2 new partial reimbursements for combination anticancer therapies that included Polivy. Partial reimbursement for combination therapy using anticancer drugs grants reimbursement to already covered drugs that are included as part of combination therapy. Previously, combination therapies were not covered by reimbursement unless they were officially added as a whole to the reimbursement criteria. The Ministry of Health and Welfare implemented this policy in May, and earlier this month, HIRA announced 35 therapies to clarify the general principles. HIRA plans to review applications for partial reimbursement for combination therapy submitted by academic societies and make additional revisions as necessary through its Cancer Disease Review Committee. As a result, two combination therapies will be added to the partial reimbursement list in July. Polivy is the first first-line treatment for DLBLC in 20 years, but it has faced difficulties in obtaining reimbursement since its approval in 2020. As a result, it remains non-reimbursed to date. The combination therapy included in the partial reimbursement criteria this time is " polatuzumab Vedotin(non-reimbursed)+rituximab, cyclophosphamide, doxorubicin, and prednisone/prednisolone (R-CHP)" for treatment-naïve adult patients with Diffuse Large B-Cell Lymphoma (DLBCL). HIRA plans to apply reimbursement for all drugs except Polivy. A combination therapy for cervical cancer that uses the immuno-oncology drug Keytruda (pembrolizumab) has also been added. As with the previous case, partial reimbursement will be provided, so all other drugs will be reimbursed excluding Keytruda and Avastin (bevacizumab). The treatments granted partial reimbursement are: pembrolizumab + paclitaxel + carboplatin ± bevacizumab and pembrolizumab + paclitaxel + cisplatin ± bevacizumab. Reimbursement will be applied to patients with recurrent, metastatic (stage IV) cervical cancer or stage IB2 or higher that meet one or more of the following conditions: ▲positive pelvic lymph nodes (pelvic LN) after surgery, ▲positive para-aortic lymph nodes (para-aortic LN) after surgery, or ▲positive parametrium after surgery (stage I or higher, palliative treatment). The reimbursement criteria also include revisions to Tier 1 and 2 anticancer drugs. HIRA explained, “When the criteria were first established in 2006, in the details regarding the application criteria and methods for reimbursement of drugs prescribed and administered to cancer patients, drugs subject to re-evaluation, orphan drugs, or drugs with potential for abuse were classified as Tier 2 drugs and was granted use within the scope of its reimbursement criteria. Tier 1 drugs were to be administered at the discretion of the doctors within the scope of the indication and general principles for anticancer therapy." "However, since the initial enactment, new clinical evidence is being updated every year due to drug price fluctuations, the listing of generic drugs and many new high-priced anticancer drugs, and the development of new drugs with new mechanisms of actions, rendering it necessary to review the old treatment guidelines (mainly the deletion of Tier 1 anticancer drugs that are highly toxic and ineffective and the reclassification of Tier 2 anticancer drugs) and recommend the use of anticancer treatments that are more clinically proven and safer for the public. In this regard, we gathered opinions from academic societies and held TFT meetings on improving the reimbursement criteria for anticancer drugs to establish a reimbursement criteria (draft) for anticancer drugs. After final discussions by the Cancer Disease Deliberation Committee, we established new reimbursement criteria for anticancer therapies that removed the classification of anticancer drugs into Tiers 1 and 2.” However still, for cancers that are difficult to establish clinical evidence due to the small number of drug options and patient population, the current system will be maintained. Detailed information on the revisions can be found on the HIRA website (System/Policy → Drug Criteria Information → Drugs and Therapies Used for Cancer).
- Opinion
- [Reporter's View] For local gov't seeking biotech diplomacy
- by Whang, byung-woo Jun 30, 2025 06:05am
- 'BIO USA,' held in Boston, U.S., is the world's largest biotech industry conference. Notably, this year's conference was attended by officials from Korea's local government. The attending officials aimed to promote local biotech clusters and seek potential investment partners. Through this event, they intended to establish a gamer-changer to their regional growth. However, some industry officials showed both anticipation and concern. Korean local governments attending this year's BIO USA strived to meditate on biotech diplomacy. For instance, Nowon District of Seoul hosted a press conference near the event location and presented plans for 'Seoul-Digital Bio City (S-DBC).' Currently, Nowon District is reportedly discussing investment opportunities with two biotech companies in South Korea to build a biotech cluster in Chong-dong Training Depot. Daejeon City, as a key bio-cluster holding local government, also sought technology transfer and joint research partnerships through MOU signings with overseas research institutions and IR events targeting investors. Furthermore, Siheung City, Gyeonggi Province, which is promoting a biotech-cluster through an investment agreement of approximately KRW 2.2 trillion with Chong Kun Dang, also visited the event. Although the enthusiasm of these local governments was evident, accomplishments have not yet been achieved. Some local governments have only just begun their steps into the pharmaceutical and bio-industry, so news of significant investment attraction or contract signings is scarce. An industry official pointed out, "While local government's interest and investment in the bio-industry are welcome. However, if the focus is solely on trying without deliveries, it will ultimately be judged as mere showmanship." Ultimately, if local governments' overseas efforts do not lead to effective investment exchanges, they may not yield tangible results despite significant time and budget expenditure. While various local governments view the pharmaceutical and biotech industries as a future growth engine and actively engage with them, not all have a favorable view. This is partly because there are precedents of bio-clusters located across the country. Currently, there are between 20-30 large and small bio-specialized complexes established in Korea, but questions remain about whether all these clusters are functioning. Recently, the focus has shifted to improving efficiency and enhancing the competitiveness of existing bio-clusters instead of merely creating new ones. Local governments also view that it's necessary to clarify their position and strengths within this larger picture, and strive to build complementary ecosystems by avoiding redundant investment or competition. The choice of local governments to invest in the biotech industry, seeing it as a key opportunity for job creation, is not necessarily deserving of criticism. However, there are negative view regarding the aspirations of some local governments to attract global big pharma's offices and R&D centers. A biotech industry official advised, "Ultimately, to attract global big pharma, clusters in South Korea must grow to a level that makes them desirable. It's not a problem that can be solved by local governments merely providing buildings and land. They must create a self-sustaining ecosystem where excellent research personnel and companies gather." Ultimately, proper direction and cooperation are needed. For the local government's efforts in Boston to yield tangible results rather than empty echoes, industry stakeholders and policymakers need to work as a unified team, drawing a larger picture, rather than pursuing individual goals, self-serving efforts by the central government, local governments, and private companies.
- Company
- 88% registered APIs imported from China or India
- by Kim, Jin-Gu Jun 27, 2025 06:04am
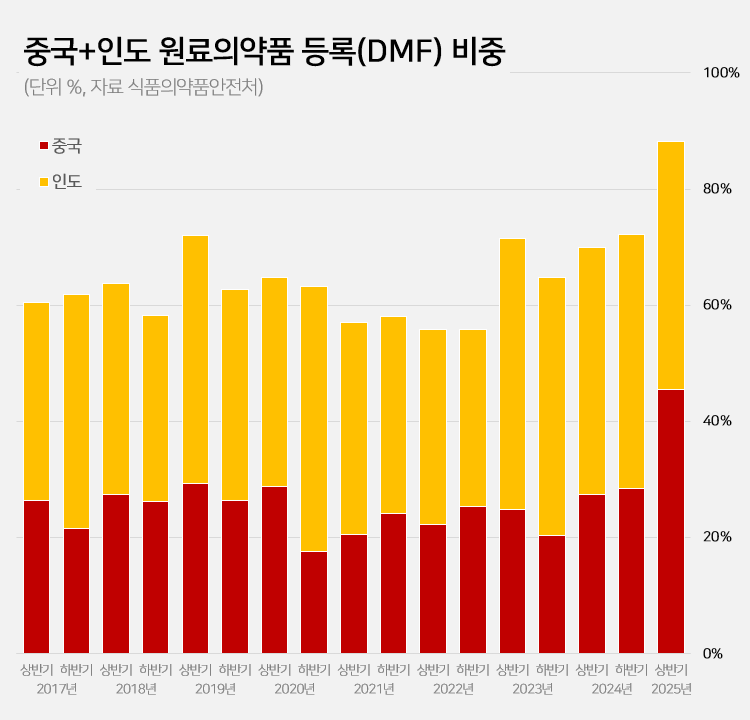
- Amid a surge in the number of drug master file registrations in the first half of this year, the share of raw materials from China and India rose to 88.2%. This is a sharp increase compared to the average of 62.1% share the two countries had during the past 5 years. This is attributed to the large number of previously delayed raw material drug registrations that had been made upon the relaxation of DMF regulations, as well as the domestic pharmaceutical and bio industry's increased use of raw materials from China and India to reduce costs. 606 DMF registration from China and India source materials made in the first half of this year... accounts for 88%, which is the highest-ever share According to the Ministry of Food and Drug Safety on the 26th, 687 DMF registrations were made in the first half of this year. Among them, 313 raw materials were from China and 293 were from India. The two countries combined accounted for 606 cases or 88.2% of the total DMF registrations. This is the highest proportion ever recorded for a half-year period. Until last year, the proportion of Chinese and Indian raw materials had never exceeded 75%. The average proportion of Chinese and Indian DMF imports over the past 5 years was 62.1%, which is more than 26 percentage points higher than in the first half of this year. The proportion of Chinese and Indian imports in the DMF has increased rapidly over the past 3 years. After steadily declining since the first half of 2019, the share of Chinese and Indian DMF rose to 55.8% in the first half of 2022 and then began to increase. It reached 69.9% in the first half of last year and 72.2% in the second half. In the first half of this year, it soared to nearly 90%. Pharmaceutical industry's dependence on Chinese and Indian raw materials deepens amid cost pressures The rapid increase in Chinese and Indian DMF is attributed to cost reduction pressures in the pharmaceutical and bio industry. China and India are representative “low-cost mass production bases” in the global raw material drug market. Following the global economic downturn after the pandemic, the pharmaceutical and bio industry in general faced a decline in profitability. As a result, attempts to reduce costs were made, which led to an increase in the use of Chinese and Indian DMF. An industry insider explained, “Domestic pharmaceutical and biotechnology companies are feeling a significant burden from manufacturing costs due to high exchange rates, rising labor costs, and declining profitability. Chinese and Indian raw materials are sometimes almost half the price of domestically produced materials, leading to increased use of imported materials.” Additionally, the relaxation of DMF regulations has further increased the use of Chinese and Indian raw materials. The government eased DMF requirements earlier this year by replacing on-site GMP inspections with the submission of GMP certificates and reducing the administrative processing period from 120 days to 20 days. As a result, imported raw materials for which registration had been delayed were registered en masse. In particular, it is analyzed that the abolition of on-site inspections has led to a significant increase in Chinese and Indian raw materials. In the past, inspections in these two countries were physically difficult, and administrative procedures complex, often causing delays in registration. This year, however, registration became possible with only a GMP certificate, significantly lowering barriers, and leading to a significant increase in the registration of Chinese and Indian raw materials. Domestic raw material share in DMF only 5%, raising concerns about increased dependence on Chinese and Indian products On the other hand, the share of domestically produced raw materials registered in the DMF has decreased significantly. In the first half of this year, the share of domestically produced raw materials registered was only 4.9% (34 cases). This is less than half of the 12.6% recorded in the second half of last year. The share of DMF registrations of raw materials from Europe and Asia also decreased sharply. The share of European raw materials decreased by more than 10 percentage points from 14.5% in the first half of last year to 4.4% in the first half of this year. The number also decreased from 37 to 30. The share of DMF registrations of raw materials from Asian countries other than China and India also decreased from 3.9% to 1.5%. Concerns have been raised that the dependence on raw materials from China and India may become excessively high. If this trend intensifies, it could pose a threat to the stability of domestic drug supply. In fact, during the early stages of the COVID-19 pandemic, export restrictions imposed by China and India directly impacted domestic drug production. A pharmaceutical industry insider stated, “If raw material production becomes overly concentrated in specific countries, it becomes vulnerable to external factors such as export restrictions, logistics disruptions, and sharp exchange rate fluctuations. In the long term, policies are needed to strengthen domestic raw material production capabilities and provide various incentives for the use of domestically produced raw materials.”
- Policy
- Ensuring stable supply of drugs in short supply
- by Lee, Jeong-Hwan Jun 27, 2025 06:03am
- Following President Lee Jae-myung's pledge to establish a stable supply system for drugs with supply shortages, attention is drawn to the Ministry of Health and Welfare's (MOHW) opinion that a social consensus on the criteria and scope of 'supply shortage' is first needed. It is anticipated that legislative review in the National Assembly to resolve the issue of drug shortages will progress if criteria for drug shortages are established, including whether the causes of the shortages are isolated or chronic. On June 26, an MOHW official met with the Korea Special Press Association and explained, "The government also agrees on the need to reform national governance to respond to drugs with supply shortages." The official proposed the necessity of establishing a definition and criteria for drugs with supply shortages to find solutions to the drug shortage problem properly. Establishing the criteria first is necessary because drug shortage issues arise from various types and cases, and finding a consensus on how to quantify them is essential for laying the groundwork to establish policies and proceed with legislation. The MOHW stated that while national essential medicines are designated through various criteria and procedures, defining criteria for drugs with supply shortages will be challenging due to their often variable nature, such as differing shortage durations for each case. An MOHW official explained, "It's crucial how we measure supply shortages. We monitor the situation through supply history reports, but it's difficult to confirm why shortages occur at the final distribution stage," and added, "Since we do not have any information on the inventory levels so there are data limitations in determining whether a shortage is due to an absolute lack of supply or a hoarding problem." The official further explained, "It takes about 2-3 months for data on actual drug prescriptions and usage to accumulate. Hospitals and pharmacies don't report every time a drug is used, and it's even harder to secure data for non-reimbursable drugs," and added, "We need to establish some level of supply shortage criteria to come up with countermeasures. Even during public-private meetings, we discussed whether a drug with supply shortage needed an intervention or not." Meanwhile, four amendments to the Pharmaceutical Affairs Act aimed at resolving the issue of drugs with supply shortages are currently pending in the National Assembly (proposed by Rep. Han Jeoung-ae, Rep. Kim Yoon, Rep. Kim Sunmin, and Rep. Seo Mi-hwa).