- LOGIN
- MemberShip
- 2026-04-03 12:18:23
- Company
- Tevimbra adds esophageal, gastric, lung cancer indications
- by Whang, byung-woo Jun 27, 2025 06:02am
- Pic of TevimbraBeiGene Korea (Name to be changed to BeOne Medicine Korea) announced that its immuno-oncology drug Tevimbra (tislelizumab) has been approved by the Ministry of Food and Drug Safety for additional indications for esophageal cancer, gastric cancer, and non-small cell lung cancer. With the additional approval, Tembriva can now be used as a first- or second-line treatment for a total of 5 indications across 3 solid tumor types. The approved cancer types are: esophageal squamous cell carcinoma (ESCC), gastric or gastroesophageal junction adenocarcinoma (G/GEJ), and non-small cell lung cancer (NSCLC). Tevimbra demonstrated efficacy and safety for the indications in the RATIONALE clinical trial series (RATIONALE-303, 304, 305, 306, 307), which served as the basis for this approval. In particular, the drugs’ clinical benefits were observed in the overall patient population for esophageal squamous cell carcinoma and gastric or gastroesophageal junction adenocarcinoma and showed consistent results in pre-specified subgroups based on PD-L1 expression levels. Such benefits of Tevimbra were also reflected in global treatment guidelines and is recommended at a high level by the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO). Tevimbra employs a dual mechanism of action that effectively blocks PD-L1 while minimizing binding to Fc-gamma receptors (FcγR), thereby inducing potent antitumor responses through a mechanism distinct from that of existing immuno-oncology agents. Also, the drug demonstrated superior PD-1/PD-L1 blocking efficacy (>99%) compared to other immunotherapy agents of the same class, and according to the company, it has a higher binding affinity and a half-life 30-80 times longer than existing drugs, suggesting a more sustained therapeutic effect. In addition, by minimizing binding to the Fc gamma (Fcγ) receptor of the antibody, the drug enhanced the sustainability and stability of the immune response. Ji-Hye Yang, General Manager of BeOne Medicines Korea, explained, “Based on its differentiated mechanism and long-term clinical data, Tevimbra is gaining attention as a new standard of care that surpasses the limitations of existing immunotherapy drugs by offering treatment performance comparable to global benchmarks, as well as treatment sustainability and financial predictability.” Yang added, “We are particularly pleased to offer new treatment opportunities for underserved patients. in the first-line setting for esophageal squamous cell carcinoma, as Tevimbra is the only approved immunotherapy in Korea that can be used regardless of PD-L1 expression levels.” Meanwhile, BeiGene changed its corporate name to BeOne Medicines under the vision of “Overcoming Cancer Together” and is accelerating the expansion of its next-generation anti-cancer portfolio, including ADC (antibody-drug conjugates) and protein degraders, with Switzerland as its global hub. The domestic subsidiary will also complete its rebranding as ‘BeOne Medicines Korea’ by June 30 and plans to accelerate its growth in the solid tumor and blood cancer treatment markets based on its 2 products and 11 indications.
- Company
- KPTA ‘KOR-CHN-JPN supply cooperation to bring $12B effect'
- by Kim, Jin-Gu Jun 27, 2025 06:02am
- The Korea Pharmaceutical Traders Association (KPTA), China Chamber of Commerce for Import & Export of Medicines & Health Products (CCMPHIE), and Japan Pharmaceutical Traders Association (JPTA) announced on June 25 that they signed a memorandum of understanding (MOU) for the stabilization of the pharmaceutical supply chain at the Korea Pavilion in the CPHI & PMEC CHINA exhibition hall in Shanghai, China. The signing ceremony was attended by Hyung-seon Ryu, Chairman of the KPTA; Zhou Hui, President of CCCMHPIE; Ichiro Fujikawa, President of JPTA; Young-soo Jeong, Director of the KOTRA Shanghai Trade Office; and over 20 representatives and officials from each country. The MOU was signed to improve the global pharmaceutical supply chain, which has become unstable following the COVID-19 pandemic, and to enhance the three countries' capability to respond to health crises through cooperation. The three associations play leading roles in the pharmaceutical trade and distribution sectors. Through the MOU, the parties agreed to collaborate on ▲ the export, import, development, and supply of essential and active pharmaceutical ingredients; ▲ exchange of pharmaceutical research personnel, technology, and information; and ▲ joint hosting of seminars, academic conferences, and workshops to promote pharmaceutical trade. They plan to concretize the outcomes of the MOU through the implementation of practical joint projects. The KPTA stated that if supply chain stabilization is realized through this agreement, it will bring an annual economic effect of approximately USD 12 billion through ▲the reduction of raw material inventory costs ▲reduction of procurement costs through joint purchasing ▲reduction of health crisis response costs ▲trade creation effects from the activation of pharmaceutical trade among the three countries ▲trade diversion effects from replacing overseas pharmaceutical imports with intra-regional trade. Also, based on this agreement, it is anticipated that a three-country contract manufacturing (CDMO) model may be forged, enabling Korean companies to produce pharmaceuticals at competitive prices in China, register them in Japan, and enter the European market. Cases where domestic pharmaceutical companies experienced disruptions in imported raw material supplies also also expected to decrease, as the partnership will allow Korean companies to secure stable supply through emergency supply contracts with Chinese and Japanese companies. Hyung-seon Ryu, Chairman of the KPTA, stated, “Korea has strengths in producing high-quality drugs, Japan in precision manufacturing technology and rare drug raw material technology, and China in large-scale production and supply capabilities. This MOU will greatly contribute to the stability of the entire Northeast Asian supply chain and the enhancement of global competitiveness. We ask Korean companies to actively participate in various areas, including follow-up technical collaboration, research and development, and contract manufacturing.” Zhou Hui, Chairman of CCCMHPIE, said, “This agreement is the result of close collaboration between the 3 countries and will contribute not only to the stable supply of essential and raw pharmaceuticals but also to the creation of new business opportunities. It will serve as a leading model for cooperation in the East Asian pharmaceutical industry.” Ichiro Fujikawa, President of the JPTA, commented, “Korea and China are Japan’s core partners for raw materials and finished pharmaceutical products. This agreement will help alleviate the shortage of pharmaceutical supplies in Northeast Asia and positively impact the growth and development of each country’s pharmaceutical industry.”
- Company
- ‘Wegovy, a game-changer for high-risk obesity patients’
- by Whang, byung-woo Jun 26, 2025 06:08am
- Obesity is a cause of various metabolic syndromes and a major risk factor for cardiovascular disease. In fact, approximately 80% of patients hospitalized for cardiovascular disease are obese, and studies have shown that the risk of cardiovascular events in obese patients is up to twice as high as in those of normal weight. Over the past 20 years, the mortality rate from obesity-related cardiovascular diseases has increased significantly, with approximately two-thirds of obesity-related deaths attributed to cardiovascular diseases. Recently, semaglutide (brand name: Wegovy), a GLP-1 receptor agonist, has opened a new treatment paradigm in obesity treatment, demonstrating efficacy in reducing the risk of major cardiovascular events in high-risk obese patients. Kim Kyung-hee, professor of cardiology at Incheon Sejong Hospital (Director of the Heart Transplant Center), met with Dailypharm and emphasized the importance of obesity treatment for the prevention of cardiovascular disease. Obesity increases the risk of early onset of cardiovascular disease... “The number of young patients is also on the rise” Obesity is a precursor to various metabolic diseases, and inflammatory substances secreted from visceral fat reduce blood vessel elasticity. According to Professor Kim, these changes lead to hypertension, diabetes, and hyperlipidemia, which in turn greatly increase the risk of early onset of coronary artery disease and heart failure even in younger age groups. Kyung-hee Kim, Professor of Cardiology, Incheon Sejong Hospital (Director of the Heart Transplant Center) Professor Kim said, “Obesity can be a cause of all cardiovascular diseases, and there is a recent trend of an increase in patients with high blood pressure or symptoms of heart failure from a young age. Recently, the prognosis of severely obese patients is generally worse than that of the general population, but even in lean individuals, and those with sufficient muscle mass tend to have a better prognosis.” Kim further explained, “In cases of cardiovascular diseases such as angina or heart failure, there is a tendency for weight loss and reduced muscle mass due to decreased appetite and nutrient intake. Therefore, obesity typically occurs first, followed by cardiovascular diseases in most cases.” In other words, obesity often acts as a precursor to cardiovascular disease. In this regard, semaglutide is regarded a game changer in the fields of obesity treatment and cardiovascular disease prevention. Semaglutide was approved by the Ministry of Food and Drug Safety in April 2024 as an anti-obesity treatment for patients with a BMI of 27 kg/m² or higher (with comorbidities) or 30 kg/m² or higher, and in July of the same year, it was additionally approved for reducing the risk of cardiovascular events in overweight and obese adult patients with confirmed cardiovascular disease. Professor Kim emphasized the clinical value of semaglutide not merely as a weight-loss aid but as a preventive therapy for cardiovascular disease. In particular, KIM highlighted findings from the SELECT trial, a pivotal clinical study on semaglutide, where the drug demonstrated an additional 20% reduction in the risk of major adverse cardiovascular events (MACE) when added to standard care in patients already receiving conventional treatments. Professor Kim explained, “In the SELECT study, approximately 90% of participants were already receiving standard treatment, but when they added semaglutide, an additional 20% reduction in MACE risk was observed. This result demonstrates that semaglutide can make a substantial contribution to improving outcomes in high-risk patient populations where existing treatments have limitations.” According to the detailed results of the SELECT trial, over an average follow-up period of approximately 3.3 years, the semaglutide 2.4 mg group showed a statistically significant 20% reduction in the risk of cardiovascular death, nonfatal myocardial infarction, or stroke compared to the placebo group. Professor Kim added, “Semaglutide regulates the appetite center in the brain, delays gastric emptying to induce weight loss, and further improves cardiovascular risk factors through its anti-inflammatory effects. While weight loss may also play a role, we believe that the drug’s anti-inflammatory effect plays a very significant role in cardiovascular health.” In particular, Professor Kim emphasized that semaglutide is not simply a weight loss drug, but a scientifically proven cardiovascular treatment option. He said, “Semaglutide is a must-use drug for patients who are severely obese or have a BMI of 27 kg/m² or higher with cardiovascular disease.” “Limitations remain on its reimbursement... Selective reimbursement support needed for high-risk obese patients” Although semaglutide has emerged as an important drug that should be considered as part of a standard treatment for obese patients at high risk of cardiovascular disease, there are currently practical limitations to its access in Korea. This is because it is not yet covered by insurance in Korea. Professor Kim said, “Drugs such as semaglutide carry a certain risk of misuse, so caution should be exercised when expanding reimbursement to all patient groups. However, I believe it is desirable to allow reimbursement through strict criteria and screening procedures for high-risk groups, such as patients with severe obesity and cardiovascular complications, for whom clear therapeutic effects can be expected.” In fact, limiting reimbursement to groups with a clear clinical need for reimbursement, such as patients with a body mass index (BMI) of 27 kg/m² or higher and obesity-related complications or cardiovascular disease, may be a realistic alternative. Professor Kim also predicted that discussions on whether to continue reimbursement will be necessary when semaglutide significantly improves a patient’s BMI. He said, "If patients with a BMI of 30 kg/m² or higher are administered semaglutide and their BMI falls below 26 kg/m² due to weight loss, it may be possible to consider limiting the reimbursement period to the initial 4-6 months. However, since there is currently insufficient long-term data in Korea and some patients experience weight regain after 6 months of treatment based on clinical experience, further review of long-term management strategies is necessary.” To address current issues surrounding reimbursement coverage and costs, Professor Kim is currently conducting an economic evaluation study. “We are analyzing how much the number of medications taken by patients can be reduced when they lose weight after 6 months or a year through bariatric surgery or semaglutide treatment, and we expect this to be significant in terms of establishing future treatment strategies and fiscal efficiency.” Ultimately, Professor Kim believes that obesity treatment should not end with medication alone but must include comprehensive management to help patients fundamentally improve their lifestyle habits. Professor Kim emphasized, “Patients with a BMI of 30 kg/m² or higher often find exercise difficult, so they should be actively educated to combine medication with walking exercises, maintain a high-quality diet, reduce carbohydrate intake, and abstain from alcohol and smoking.” “Early intervention in obesity treatment is necessary to maximize preventive effects” Professional counseling and lifestyle education support are essential to increase the effectiveness of obesity treatment. However, the reality of how difficult it is to provide sufficient counseling in the current outpatient setting is also pointed out as an issue. Professor Kim said, “It is difficult to check blood pressure, assess the patient's condition, perform a physical examination, and explain lifestyle correction measures within the 5 minutes of consultation time allocated per patient. At least 7-10 minutes are necessary for proper treatment.” In this regard, Professor Kim proposed the establishment of a lifestyle education program and a new fee schedule to hire dedicated personnel to overcome such limitations. He explained, “Under the current system, separate reimbursement rates for education provided by specialized nurses need to be introduced, and institutional and financial support would also be needed to manage these personnel. Overall, I believe that establishing an environment where lifestyle education can be systematically implemented is essential to improving the quality of care for obese patients. In particular, Professor Kim emphasized, “Patients with hypertension are at high risk of developing heart failure over time. Starting medication early and educating young obese patients with hypertension on proper lifestyle habits can prevent serious complications and repeated hospitalizations.” In other words, Kim believes preventive treatment and lifestyle improvement efforts targeting young obese patients are expected to lead to macro-level medical cost savings in the future. Finally, Professor Kim emphasized that “Patients should always be treated with scientifically validated medications first. Drug treatment alone is not sufficient and must be accompanied by lifestyle modifications and proper dietary management.”
- Company
- The 2nd KRAS-targeted cancer drug 'Krazati' expected
- by Eo, Yun-Ho Jun 26, 2025 06:08am
- Product photo of Krazati The second KRAS inhibitor is expected to be commercialized in South Korea. Bristol Myers Squibb (BMS) Korea recently submitted a marketing authorization application to the Ministry of Food and Drug Safety (MFDS) for its anti-cancer drug, Krazati (adagrasib). Krazati was also designated as an orphan drug in January. It is indicated for the treatment of 'locally advanced or metastatic non-small cell lung cancer (NSCLC) with a KRAS G12C mutation, previously treated with at least one prior therapy.' Krazati received accelerated approval from the U.S. FDA in December 2022. It is the second KRAS inhibitor receiving approval, following Amgen's 'Lumakras (sotorasib),' which was approved in 2021. The development of KRAS-targeted anti-cancer drugs has come approximately 40 years after the initial discovery of the oncogene. Amgen and BMS are engaged in fierce competition to dominate this new market. Lumakras and Krazati share many similarities, including their target mutation and indications. Both target the KRAS G12C mutation, and their initial approved indication is NSCLC. Both companies are also conducting clinical trials in combination with compounds with different mechanisms of action developed in-house or through collaborations. Meanwhile, Krazati's initial approval was based on cohorts from the KRYSTAL-1 study who are eligible for Phase 2 trials. Last year, the primary analysis results of the confirmatory Phase 3 study were disclosed. This study compared Krazati with docetaxel in 301 patients with previously treated KRAS G12C-mutated locally advanced or metastatic NSCLC. These patients had previously received platinum-based chemotherapy and anti-PD-1/PD-L1 immunotherapy. They were randomized 1:1 to either the Krazati treatment group or the docetaxel treatment group. The primary endpoint was progression-free survival (PFS) as assessed by blinded independent central review (BICR). After 9.4 months of follow-up, the median PFS for Krazati was 5.49 months, which met the primary endpoint by reducing the risk of disease progression or death by 42% compared to 3.84 months for the docetaxel group.
- Product
- 'Govn’t must take strong action against pharma rebates'
- by Kang, Hye-Kyung Jun 26, 2025 06:08am
- The Korean Pharmacists for Democratic Society (President Gyeong-rim Jeon, KPDS) has urged the government to take strong action against pharmaceutical company rebates. KPDS issued a statement regarding rebates made by a major domestic pharmaceutical company that JTBC reported. In the statement, KPDS said, “This is not an individual incident, but clear evidence of widespread illegal rebates rampant throughout the pharmaceutical industry. “Medicines prescribed by doctors are not ordinary commodities, but special goods that directly affect patients' lives and health. The Pharmaceutical Affairs Act and Medical Service Act fundamentally prohibit pharmaceutical companies from providing economic benefits to medical professionals.” However, according to the JTBC report, the pharmaceutical company in question blatantly violated legal restrictions. The KPDS criticized, “What is even more outrageous is the government authorities' incompetent response to these clear illegal acts. Using a shortage of personnel as an excuse to avoid addressing the issue raises suspicions that there may be an intent to cover up the incident.” They went on to say, “The explanation that this was a legitimate new drug promotion activity is a clear lie, and the government must immediately implement strong measures to eradicate illegal rebates.” In other words, the KPDS believes the MOHW and the police must reinvestigate the case and severely punish those involved. The KPDS proposed the following measures: ▲reinvestigation through the formation of a special investigation team; ▲a comprehensive investigation by the MOHW into the provision of financial benefits such as support for academic conferences, lecture fees, and consulting fees; and ▲strong administrative measures such as drug price reductions when illegal rebates are detected. They emphasized, “We can no longer tolerate the illegal rebate cartel that threatens the health insurance budget and public health. The government must use this incident as an opportunity to demonstrate its firm commitment to eradicating illegal rebates in the pharmaceutical industry.”
- Policy
- Yungjin, Ildong’s Ofev generics enter market at half price
- by Lee, Tak-Sun Jun 26, 2025 06:07am
- A generic version of Ofev (nintedanib), a treatment for chronic fibrotic interstitial lung disease), will enter the market at half the price of the original drug. With the entry of generic drugs, the Ofev market now faces competition, just two months after the original drug was listed for reimbursement. According to industry sources on the 25th, Yungjin and Ildong Pharmacuetical will list the generic version of Ofev with reimbursement at a price much lower than the calculated price, differentiating it from the original product. Also, the original product is a capsule formulation, while the generic version is a tablet formulation. When reimbursement was approved in May, the maximum insurance price for Boehringer Ingelheim Korea's Ofev Soft Capsules 100 mg was set at KRW 29,600, and Ofev Soft Capsules 150 mg at KRW 26,220. The generic versions are much cheaper than this. Three generic drugs will be listed for reimbursement next month: Yungjin Pharmaceutical's Nintebro Tab 150 mg and Nintebro 100 Tab mg, and Ildong Pharmaceutical's Cuninta Tab 150 mg. Among them, Nintebro 100 mg has a maximum price of KRW 9,000, less than half the original Ofev Soft Capsule 100 mg (KRW 20,960). Nintebro 150 mg is KRW 15,000, and Cuninta 150mg is KRW 13,500. When considering how Ofev Soft Cap 150mg costs KRW 26,220, the generic versions cost half the original price. Being rare disease treatments, the generic versions were eligible to receive the same price as the original Ofev, but it is believed that the companies significantly lowered the price in consideration of market competitiveness. As a result, patients will now be able to purchase the same ingredient drug at a lower price. In May, after nine years since being approved in Korea, Ofev was listed for reimbursement as a treatment for chronic fibrotic interstitial lung disease. Due to the delay in reimbursement listing, the substance patent expired on January 25. This is how generic versions were able to enter only 2 months after the original drug was listed for reimbursement. Meanwhile, Yungjin and Ildong, which are launching the generic versions, are competing fiercely in the pirfenidone market, a representative treatment for idiopathic pulmonary fibrosis, which is another indication for Ofev.
- Company
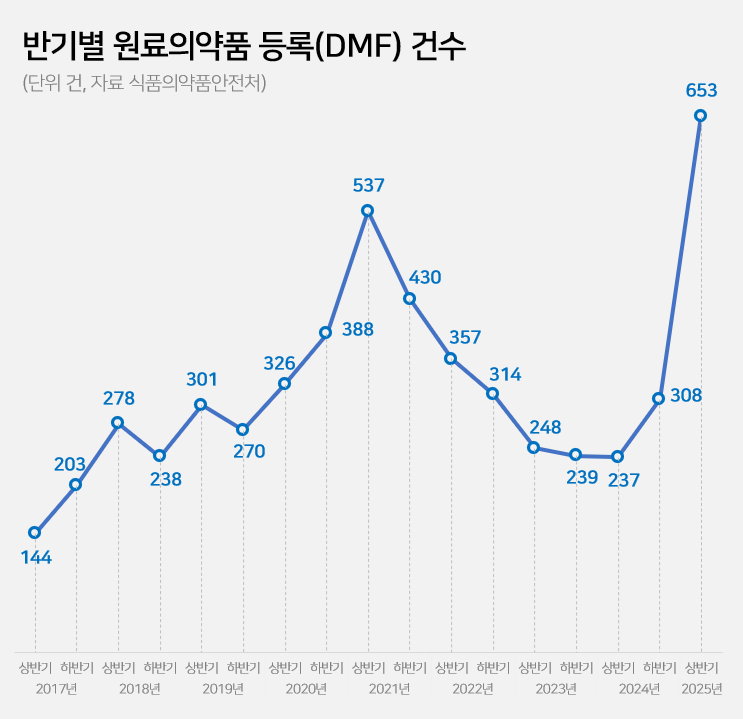
- Drug Master File for API 237→653…'easing of regulation'
- by Kim, Jin-Gu Jun 26, 2025 06:07am
- The number of Drug Master File, DMF, cases in the first half of this year surged by 2.8 times compared to the same period last year. This is the highest for a half-year period. Analysis suggests that this is due to the easing of Active Pharmaceutical Ingredient (API) registration requirements at the beginning of the year. The government had previously eased regulations to allow GMP evaluation to be replaced by GMP certificates for API registration starting this year. 'Record-High' DMF Registrations of 653 Cases in the first half of 2025...Up 2.8x YoY According to the Ministry of Food and Drug Safety (MFDS) on June 26, the number of API registrations by Korean pharmaceutical and biotech companies in the first half of this year reached 653 cases. Compared to 256 cases in the first half of last year, this year's marks a 2.8-fold increase in one year. It has already surpassed the total number of API registrations for the entire 2024 (545 cases). In terms of half-yearly API registrations, it has exceeded the 537 cases in the first half of 2021, reaching an all-time high. Half Yearly Cases of Drug Master Files (DMFs) for API (unit: number of cases, source: MFDS) The robust increase in DMF cases is attributed to the government's easing of regulations. Earlier this year, MFDS reformed the DMF system to replace on-site inspections with GMP certificates. Previously, DMF applications required on-site inspections, along with manufacturing facility data, production country manufacturing certificates, and 11 types of GMP documents. From this year, on-site inspections have been abolished. Additionally, documents can now be replaced by GMP certificates issued by the government agency of the production country or a PIC/S member country. The administrative processing period has also been shortened from 120 days to 20 days. An MFDS official explained, "Previously, to register API, the applying company had to undergo a GMP on-site inspection, but from this year, it can be substituted with a certificate," and added, "It seems that nearly 1,000 piled-up DMF applications were processed in large numbers this year, leading to a surge in DMF cases." Concerns over API quality verification...MFDS states "On-site inspections maintained for high-risk Items" Regarding this deregulation, some in the pharmaceutical industry express concerns that API quality control could become lax. Critics argue that, with registration now possible solely based on GMP certificates, it will be challenging to identify quality issues beforehand through document-based evaluations. This easing of regulation is a complete reversal from MFDS's previous stance. Since the introduction of the DMF system in 2002, MFDS has consistently strengthened quality control. In 2014, GMP evaluation standards were reinforced with PIC/S membership. At this time, 11 types of GMP documents and on-site inspection standards were introduced. In 2019, DMF registration became mandatory not only for new items but also for previously approved items. In 2021, the on-site inspection system was further strength with a focussing on high-risk items. During a briefing last year, MFDS explained that they adjusted the evaluation system in response to the administrative bottleneck caused by a surge in DMF applications, which also delayed the review of finished pharmaceutical products (FPP). Overall, MFDS's policy is to shift its GMP approach to be 'FPP-centric.' Regarding concerns about API quality, MFDS states that on-site inspections are exceptionally maintained for high-risk items, and GMP certificate requirements have been strictly set in line with international standards. Indeed, for high-risk items such as biopharmaceuticals and sterile APIs, on-site inspections and submission of evaluation data are still required. Furthermore, on-site inspections are maintained as before for drug approval and suitability judgments. They also plan to introduce the concept of a 'Site Master File' to understand the quality management system of manufacturing sites comprehensively. Up and down of cases based on regulatory changes...Decline after 2021 peak→ rebounding This Year The number of DMF registrations by Korean pharmaceutical and bio-companies has fluctuated significantly each year due to system changes and policy factors. Over the past eight years, DMF cases have exhibited fluctuating trends: ▲347 in 2017 ▲516 in 2018 ▲571 in 2019 ▲714 in 2020 ▲967 in 2021 ▲671 in 2022 ▲487 in 2023 ▲545 in 2024. With 653 cases in the first half of this year alone, there is a possibility of exceeding 1,000 cases by year-end. The surge in DMF cases in 2021 coincided with the implementation of a policy that made API registration mandatory, even for previously approved items. In 2019, MFDS expanded the scope of DMF to include 'previously approved items' from the original 'newly approved items.' It is analyzed that commercial drugs were required to complete registration by 2021, leading to a concentrated influx of DMF applications. A reform of the drug pricing system around the same time also influenced the increase in DMFs. In July 2019, the government introduced a 'step-wise drug pricing system.' Generics that did not meet the highest price criteria could maintain their previous drug prices if they submitted data from bioequivalence tests and demonstrated the use of registered APIs. This led to a surge in DMF applications from pharmaceutical and biotech companies seeking to maintain drug prices. After 2023, the situation changed. In February 2023, the submission of DMF documents for drug price maintenance concluded. With most DMFs for previously approved items also finalized, the number of registrations began to decline. Indeed, DMFs, which had reached 967 cases in 2021, nearly halved to 487 cases by 2023. However, with the lowering of DMF hurdles this year, the number of registrations is showing a rebounding trend.
- Policy
- Daewoong's high-dose generic 'Xeljanz' wins nod
- by Lee, Hye-Kyung Jun 26, 2025 06:06am
- Product photo of Daewoong Pharmaceutical's oral generic version of 'Xeljanz (tofacitinib),' which is used to treat rheumatoid arthritis, received approval. On June 23, the Ministry of Food and Drug Safety (MFDS) approved Daewoong Pharmaceutical's two dosages of 'Xeltofa Tab': 5 mg and 10 mg. The latest approval grabs attention, particularly because Daewoong Pharmaceutical has developed high-dose formulation of generic Xeljanz. Over 65 generic Xeljanz have been approved to date. However, only five items with a 10 mg high-dose formulation, including Pfizer Korea's 'Xeljanz,' Chong Kun Dang's 'Tofacell Tab,' Jeil Pharm's 'Topazan Tab,' Jeil Pharm's 'Topazan Tab,' and Alvogen Korea's 'Gencinib Tab,' are available. High-dose formulations have not been popular because the approved indications were fewer than those of low-dose ones. While the low-dose formulation holds five indications, including rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, ulcerative colitis, polyarticular juvenile idiopathic arthritis (pJIA), and juvenile psoriatic arthritis, the high-dose formulation is only approved for ulcerative colitis, thereby limiting its therapeutic scope. Safety concerns and fewer indications have hindered high-dose generic Xeljanz approvals. However, an increasing number of pharmaceutical companies are now securing approvals for both low-dose and high-dose products. With the substance patent for Xeljanz expiring in November, Korean companies are expected to expedite getting approvals for their generics. Xeljanz holds a substance patent expiring in November 2025 and a crystal form patent expiring in November 2027. Twenty companies, initially by Chong Kun Dang, attempted to overcome these patents by filing invalidation trials. Additionally, 16 companies, including Boryung Pharmaceutical, filed passive rights scope confirmation trials to circumvent them. In January 2018, generic companies successfully circumvented the Xeljanz patent. Following another victory in an invalidation trial in November 2019, the Xeljanz crystal form patent was removed in March 2020. Generic Xeljanz items that have obtained marketing authorization can be launched after November 22, 2025, when the substance patent expires.
- Opinion
- [Reporter's View] Silent efforts pay off in the market
- by Son, Hyung Min Jun 25, 2025 06:02am
- In the first half of this year, South Korean pharmaceutical and biotech companies achieved notable success through technology exports. A series of major licensing deals were signed based on the companies’ innovative drug development platforms and clinical trial results the companies steadily developed and accumulated over the years. In April, ABL Bio signed a technology licensing agreement with GlaxoSmithKline (GSK) for its blood-brain barrier (BBB) shuttle platform, ‘Grabody-B,’ for the development of new treatments for neurodegenerative diseases. Through this deal, ABL Bio received approximately KRW 73.9 billion as a non-refundable payment. The total contract value is estimated at around KRW 4 trillion. This is ABL Bio's seventh technology export deal. Previously, the company had successfully licensed its technology to major pharmaceutical companies such as Sanofi and Yuhan Corp. Alteogen has transferred its subcutaneous (SC) formulation change platform ‘ALT-B4’ to MedImmune, a subsidiary of AstraZeneca. Alteogen has signed two licensing-out deals with MedImmune's US and UK subsidiaries. The two contracts are worth KRW 65.5 billion, with the total contract value, including milestone achievements, exceeding KRW 2 trillion. Alteogen has also signed licensing deals with leading global pharmaceutical companies such as MSD and Daiichi Sankyo. Through the agreements, the company’s formulation change technology will be applied to major immuno-oncology drugs like Keytruda and the antibody-drug conjugate (ADC) Enhertu. In February, Olix Pharmaceuticals successfully exported its technology for OLX75016, a new drug candidate for obesity and metabolic dysfunction-associated steatohepatitis (MASH), to Eli Lilly. OLX75016 is a MASH and obesity treatment candidate based on siRNA technology, a short double-stranded RNA genetic material that plays a key role in RNA interference. OLX75016 is being developed as a subcutaneous (SC) formulation obesity treatment that can be administered once every three months. The common thread among the three companies lies in their long-standing pursuit of innovative drug development platform technologies. ABL Bio's blood-brain barrier (BBB) shuttle technology, Alteogen’s SC formulation conversion technology for anticancer drugs, and Olix's siRNA platform for obesity treatments are the result of repeated animal studies and early-phase clinical trials—achievements compelling global pharmaceutical companies to initiate deals. The upfront payments alone amount to hundreds of billions of won, with total contract values exceeding the trillion-won mark. These technology export cases are not simple pipeline transactions. It holds significance in that global pharmaceutical companies are knocking on Korean companies’ doors, recognizing the platform itself as a technological asset. Platform technology is not limited to a single substance but can be expanded to the development of multiple candidate substances, which is an option that reduces long-term risk for partner companies. In particular, these achievements are not one-time contracts. They have great potential to lead to subsequent pipeline expansion, joint development, and long-term commercialization partnerships. This demonstrates the power of a ‘technology platform’ that goes beyond simply selling a single technology that enables a future together with partners. Developing a model that can prompt a series of technology exports to multiple partners based on a single platform is the direction that the domestic pharmaceutical and bio industry should seek. It is time to move away from licensing out just a single pipeline and adopt a platform-based approach that has scalability and repeatability. Once again, the recent licensing out deals have shown how technology with ‘sustainability’ and ‘consistency’ rather than those that chase the latest R&D trends will ultimately be chosen by the market. Clinically validated technologies, rather than optimistic projections for investor appeal, are increasingly emerging as the true standard of competitiveness on the global stage.
- Policy
- Samsung Bioepis joins Prolia market with Obodence
- by Lee, Tak-Sun Jun 25, 2025 06:02am
- With Samsung Bioepis entering the biosimilar market for Prolia (denosumab), competition is expected to intensify among co-promoter companies Daewoong Pharmaceutical (Stoboclo, Celltrion), Hanmi Pharmaceutical (Obodence, Samsung Bioepis), and Chong Kun Dang (original Prolia, Amgen). Chong Kun Dang signed a co-promotion agreement with the original manufacturer, Amgen; Daewoong Pharmaceutical with Celltrion; and Hanmi Pharmaceutical with Samsung Bioepis, respectively. According to industry sources on the 24th, Samsung Bioepis' Prolia biosimilar Obodence Prefilled Syringe (denosumab, 60mg/1mL) will be listed for reimbursement in July. As a result, Celltrion's ‘Stoboclo Pre-filled Syringe’ and Samsung Bioepis' ‘Obodence Pre-filled Syringe’ are expected to compete fiercely in the domestic market. Park Jae-Hyun, President of Hanmi Pharmaceutical (right), and Kim Kyung-Ah, CEO of Samsung Bioepis (left), sign a copromotion agreement in March and pose for a commemorative photo. Celltrion’s Stoboclo was listed for reimbursement in March. The maximum reimbursement ceiling price for Stoboclo and Obodence is the same. Obodence’s price was set the same as the lowest price of the same ingredient drug already listed when it was added to the reimbursement list. As there are between 2 and 19 drugs of the same type, and the price of the drugs have already been adjusted to 70%, Obodence’s price was calculated at the same price as the lowest price of the same drug already listed in accordance with the calculation rules for biological drugs. The lowest price among the same-ingredient drugs was Stoboclo. Stoboclo’s price was voluntarily reduced by Celltrion from KRW 111,384 to KRW 108,290 starting in July, and Obodence’s price was also set accordingly at KRW 108,290. The original Prolia’s insurance ceiling price is KRW 123,760. Prolia is the absolute leader in the osteoporosis treatment market. According to IQVIA, Prolia recorded sales of KRW 174.9 billion in the domestic market last year. Prolia suppresses the activity of osteoclasts responsible for bone degradation, thereby preventing bone resorption while promoting an increase in bone mineral density. The drug is regarded to have improved the convenience of administration for patients, as its efficacy can be maintained for 6 months with a single injection. Daewoong Pharmaceutical and Celltrion Pharmaceutical announced in October last year that they signed an agreement for the copromotion of a Prolia biosimilar With a domestic market size worth KRW 170 billion for that single drug, large pharmaceutical companies have actively joined in the competition to become the distributors of denosumab and its biosimilars. The original Prolia has been sold jointly by Chong Kun Dang and Amgen since 2017. Daewoong Pharmaceutical stepped in as a relief pitcher for Celltrion's Stoboclo. Chang-Jae Lee, CEO of Daewoong Pharmaceutical, said after the copartnership agreement signing ceremony, “We expect that the agreement will leverage the strong sales and marketing capabilities of Daewoong Pharmaceutical and the outstanding biosimilar technology of Celltrion to dominate the osteoporosis treatment market. Furthermore, we will strive to reduce the risk of fractures and improve the quality of life for osteoporosis patients in Korea with CT-P41, in a prescription market worth KRW 170 billion.” Hanmi Pharmaceutical will co-market Samsung Bioepis' Obodence. Jae-Hyun Park, President of Hanmi Pharmaceutical, said, “Hanmi Pharmaceutical is a well-prepared partner with an established foothold in the musculoskeletal treatment market. Through the partnership, we will closely collaborate with our partner to create innovative results that promote mutual growth.”