- LOGIN
- MemberShip
- 2026-04-04 16:37:08
- Company
- K-Phama companies are targeting Southeast Asia
- by Heo, sung-kyu Sep 27, 2024 05:53am
- Korean pharmaceutical companies target Indonesia to establish a bridgehead for Southeast Asia market entry. Companies have invested in Indonesia by buidling manufacturing plants. The government and associations are also in support. According to pharmaceutical companies on September 20th, pharmaceutical companies, the government, and organizations are quickly establishing collaborative partnerships with Indonesia. Recently, public-private representatives, including KPBMA, Ministry of Food and Drug Safety (MFDS), KIMCo, and 15 Korean pharmaceutical and biotech companies, have visited Jakarta, Indonesia, to seek collaborative business opportunities with Indonesian Food and Drug Authority (BPOM) and local companies. The collaboration between pharmaceutical companies and the government is critical because the industry has shown interest in entering the Indonesian market. Korean pharmaceuticals are showing intersts due the growth in Indonesia market and government's support for in-house pharmaceutical development. Indonesia's pharmaceutical and biotech market is expected to grow quickly from KRW 13 trillion in 2022 to KRW 18 trillion in 2026. The analysis suggests that population growth and increased intractable diseases, including cancer and degenerative brain diseases, from an aging society will contribute to such development. Furthermore, Indonesia is considered a hub for pharmaceutical R&D·production·consumption. It is the biggest pharmaceutical consumption market among the countries in the Association of Southeast Nations (ASEAN). Additionally, to target the "Halal Belt," which consists of approximately 1.9 billion people worldwide due to population growth in Muslim countries, the Indonesian market is considered as a bridgehead in response to the increasing demand for "Halal certification" for pharmaceuticals. For these reasons, Korean pharmaceutical companies are currently establishing joint-subsidiaries with local companies, building manufacturing plants, and out-licensing technologies, in addition to exports. In particular, countries that have already entered the market are experiencing the results. Korean pharmaceutical companies that have entered the market include Daewoong Pharmaceutical, Chong Kun Dang, GC Biopharma, SK Plasma, HK inno.N, Il Dong Pharmaceutical, and Daewon Pharm. For Daewoong Pharmaceutical, the stem cell factory in the Cikarang Javabeka Industrial Complex of Daewoong Biologics Indonesia, an Indonesian subsidiary, obtained GMP certification from the Indonesian BPOM and began full-scale operation. Since establishing a branch in Jakarta in 2005, Daewoong Pharmaceutical has steadily strengthened cooperation, including establishing a joint venture with Infion, Daewoong Infion, in 2012. These efforts are bearing fruit. Chong Kun Dang established Indonesia's first halal-certified anticancer drug production plant in 2019 after establishing a joint venture, 'CKD-OTTO,' with the Indonesian pharmaceutical company OTTO in 2015. In addition, the Indonesian joint venture is achieving success by exporting products from Indonesia to Algeria and other countries. GC Biopharma received final approval for the business license for blood product plant construction and technology transfer from the Indonesian BPOM, on June 1, 2023. The company works with the Indonesian Red Cross and local pharmaceutical company P.T. Triman to supply plasma for blood product processing and plant business. A three-party business agreement has been signed concerning this. GC Cell, an affiliate of GC Biopharma, has signed a partnership agreement with the Indonesian company 'Kalbe Farma' and entered into a technology transfer and out-licensing agreement for 'Immune Cell LCD', a patient blood-derived immune anti-cancer cell therapy. The launch is aimed for next year. SK Plasma received approval from the Indonesian BPOM in March last year to build a plasma fractionation plant that can process 1 million liters of raw plasma annually. The company plans to establish a factory in collaboration with the 'Indonesia Investment Authority (INA)' and take charge of factory operations, business rights, production, and sales. In addition to companies establishing factories, HK inno.N exports K-CAB, Daewon Pharm exports Pelubi CR tab, and Boryung exports Kanarb tab. The government also continues to provide support. On September 11th, the MFDS dispatched a pharmaceutical entry support team through a public-private partnership. The MFDS visited Korean pharmaceutical companies operating in Indonesia to discuss the current status and outlook of the Indonesian market, learn about their experience in the local market, and collect information on other difficulties. The effort to enter the Indonesian market will continue through collaboration among government bodies, such as the MFDS, organizations, and pharmaceutical companies.
- Company
- 'Treatment options for psoriasis are evolving'
- by Son, Hyung Min Sep 27, 2024 05:53am
- Dr. April W. Armstrong, Professor and Chief of Dermatology at the University of California Los Angeles (UCLA) David Geffen School of Medicine “In the field of psoriasis, the development of many biologics has been largely abandoned due to the lack of convincing data showing similar efficacy to marketed drugs. On the other hand, in the field of oral drugs, interest in the TYK2 mechanism has increased upon the introduction of Sotyktu, increasing the industry’s R&D on such oral drugs. I expect to see an active oral psoriasis treatment option development in the coming years.” Dr. April Armstrong, a professor at the David Geffen School of Medicine at UCLA, recently spoke to Dailypharm about her rising expectations in the future of psoriasis treatment upon the introduction of various psoriasis treatment options. Professor Armstrong is an expert in the field of psoriasis, having served as chair of the Medical Board at the National Psoriasis Foundation and co-president of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA). Psoriasis is a chronic, non-contagious skin condition with flare-ups and remissions, with an estimated prevalence of 3% in Korea and an estimated 1.5 million patients. It is characterized by narrow, rice-like red patches on the skin, covered with white dead skin cells, which can grow from the size of a coin to the size of the palm of your hand when the rash becomes aggravated. Psoriasis is categorized by clinical type, and in “plaque psoriasis, which accounts for 80 to 90 percent of all cases, the rash is shaped like a plaque. Psoriasis is not just a skin disease, but a systemic and persistent immunologic abnormality that is difficult to cure and requires long-term management. The goal of treatment for psoriasis is to improve the quality of life by improving skin lesions and controlling flare-ups and exacerbations while minimizing the side effects of medications and preventing the many systemic complications caused by chronic inflammation. A variety of treatment options have already emerged for psoriasis, including TNF-α inhibitors and interleukin (IL)-inhibitors. In addition, new oral therapies, such as BMS' Sotyktu, have emerged to target both therapeutic efficacy and patient convenience. Sotyktu is the first TYK2 inhibitor approved for moderate-to-severe plaque psoriasis in adults. Sotyktu is taken orally once daily, regardless of meals, with no dosage adjustments, expanding the patients’ options in a setting where biologics were the only treatment option available outside of the existing universal treatment. Professor Armstrong emphasized that Sotyktu brought the psoriasis treatment environment a step forward and that oral formulations could be the next big R&D trend. Introductino of Sotyktu...first oral treatment option to target TKY2 Sotyktu is a TYK2 inhibitor. TYK2 is an important link in the signaling pathway of IL-23, a cytokine known to play a key role in the pathogenesis of psoriasis. TYK2 mediates multiple cytokine pathways, such as IL-23, which in turn triggers the production of pro-inflammatory cytokines including IL-17 that promote keratinocyte proliferation and epidermal overgrowth, resulting in psoriasis. As a TYK2 inhibitor, Sotyktu selectively binds to the regulatory domain of TYK2 and blocks IL-23 signaling, thereby reducing the production of IL-23 and IL-17, which in turn reduces keratinocyte proliferation. “Patients with psoriasis have long suffered from a lack of adequate treatments, and only recently have started gaining access to moderately effective treatments,” said Professor Armstrong. However, there is still an unmet need. Healthcare providers need to ensure that patients have access to treatments that fit their preferences and lifestyle. Some patients may be uncomfortable with the idea of injections. Some patients avoid Injectable medications due to significant anxiety, such as fear of needles, in which case oral medications can be an alternative.” The introduction of Sotyktu expanded options for patients who have primarily been prescribed injectables. In addition to the clinical trial that became the basis of its approval, Sotyktu’s efficacy and safety were confirmed in recently published 4-year patient follow-up results. The study, named POETYK PSO-1,2, LTE, looked at the Psoriasis Area and Severity Index (PASI) 75 response rate and static Physician's Global Assessment (sPGA) 0/1 response rate from 52 weeks to 4 years in 513 patients who continuously received Sotyktu for 4 years. Results showed that 71% of patients treated with Sotyktu maintained PASI 75 over the 4 years, and 57% of patients achieved and maintained a high standard of “clear” and “almost clear” skin. “No new adverse events or signals were identified in the safety data collected over the 4 years,” said Professor Armstrong. The safety profile and tolerability seen in the parent study were well maintained in the Sotyktu arm.” “In terms of safety, Sotyktu was free of serious or major adverse events, including major adverse cardiovascular events, cancer, and venous thromboembolism. Also, the serious adverse events that may arise with the Janus kinase (JAK) 1/2/3 inhibitors, such as serious infections and psoriatic arthritis, were not reported with the use of Sotyktu.” Another strength of Sotyktu is that it is effective in Asian patients. In the PSO-3 study, which focused on Asian patients from Korea, China, and Taiwan, the PASI response rate was approximately 10% higher than that identified in the global study. “The superior data of Sotyktu identified in the Asian study may be due to the fact that Asians tend to weigh less than Westerners, as well as genetic differences,” explained Professor Armstrong. “The PSO-4 clinical data in Japanese patients showed a 76% PASI 75 rate and 83% sPGA 0/1 rate at Week 16.” “However, it should be noted that both PSO-3 and 4 trials were relatively small studies compared to global studies, and PSO-4 was a single-arm study in Japanese patients. Single-arm studies without control arms tend to show higher rates,” added Professor Armstrong. Increased psoriasis treatment options... broaden choices for patients Treatment options for psoriasis have expanded with the introduction of a variety of new drugs, including injectable and oral therapies. Various treatment options, which include IL-17, IL-23, TNF-α inhibitors, and Sotyktu, are all improving the quality of life for patients with psoriasis. However, Professor Armstrong suggests that the future trend in R&D for psoriasis treatments will likely shift toward oral agents. “With the introduction of Sotyktu, the development of biologics (injectables) in the treatment of psoriasis has gradually faded away,” said Professor Armstrong, ”Fewer new biologics are being developed, except for those that are nearing the end of development or have already been launched in other countries and are expected to be introduced to Korea.” According to Professor Armstrong, some biologics are under clinical trials in the U.S., but many of them have been discontinued due to the lack of convincing data, showing similar efficacy to drugs already on the market. In the case of IL-17 inhibitors, concerns have been raised about the exacerbation of inflammatory bowel diseases such as ulcerative colitis and Crohn's disease, as well as oral candidiasis. IL-23 inhibitors have not been associated with any significant adverse events, but there have been concerns about pain or discomfort at the injection site and upper respiratory tract infections. On the other hand, the safety of oral Sotyktu is considered to be well-established with 4 years of data. While other oral therapies have been associated with higher rates of adverse events such as nausea and vomiting than placebo, Sotyktu has been well tolerated, with rates of these events not significantly different from placebo, said Professor Armstrong. “This is an exciting time for patients with psoriasis, due to the increasing treatment options available. Contrary to how patients had to choose between safety or efficacy when opting to use oral treatment options, oral therapies have evolved since then. New oral drugs such as Sotyktu have demonstrated long-term efficacy and safety comparable to first-generation biologics such as TNF-α inhibitors and IL-12/23 inhibitors,” said Professor Armstrong. ”The treatment options have significantly improved than in the past.”
- Company
- KRPIA expresses concerns over drug approval fee hike
- by Eo, Yun-Ho Sep 27, 2024 05:53am
- “The pharmaceutical industry is bound to feel burdened by the sudden decision. We hope that the new drug approval system will be adjusted through discussions with the industry.” The Korean Research-based Pharmaceutical Industry Association (KRPIA) expressed the pharmaceutical industry's stance regarding the amendment to the 'Fee Regulations for Approval of Drugs, etc.' that was announced by the Ministry of Food and Drug Safety (MFDS) on the 9th. As part of the ‘Measure for Innovation in New Drug Approval', the MFDS is expected to dramatically increase the fee for new drug approval from KRW 8.83 million to KRW 410 million, which is a nearly a 50-fold increase, by fully applying the benefit principle. In essence, KRPIA agrees with the MFDS’s proposal, including the need to realize new drug approval fees, strengthen review capabilities, and shorten the approval period. KRPIA saw that the MFDS’ decision to significantly increase the approval fee reflects the authorities’ intention to innovate the new drug approval process to respond more quickly and flexibly to environmental changes and meet new industrial demand. However, it added the industry’s concerns regarding the fee burden, which rose significantly - over 50 times - with the sudden announcement of the revision without a grace period or phased application. As this is an unprecedented increase in the fee, the KRPIA’s position is that an approval system and administrative services should be prepared at a level that everyone can agree on through sufficient discussions with the industry. “The license fee of KRW 410 million is very high compared to almost all developed countries except the U.S. and Europe,” KRPIA said, noting that Korea's market size is only one-fourth and drug prices are only 60% of Japan's, which also has a similar fee level. With many countries racing to introduce new drugs quickly to improve patient access to treatment, Korea's pharmaceutical market size, challenging drug price environment, and Korea-specific approval requirements suggest that an excessive approval fee hike could be another factor that can slow the introduction of innovative new drugs with low prevalence or small market size. “Its implementation in January 2025 may be too soon for pharmaceutical companies to prepare for changes, and seems to be an insufficient time for the MFDS to recruit specialized personnel and overhaul the system,” said KRPIA. “In order for the intention of the system to be well realized, the fee hike must be accompanied by an overhaul of the new drug approval system and the introduction of fast and advanced administrative services," emphasized KRPIA.
- Policy
- COVID-19 drugs to be reimbursable, "KRW 50,000 co-payment"
- by Lee, Jeong-Hwan Sep 27, 2024 05:53am
- The National Health Insurance will cover two COVID-19 treatments, Paxlovid and Veklury, starting on October 1st. The government will list COVID-19 treatments for reimbursement and make a policy revision to support the patient co-payment at the previous price of KRW 50,000. The National Health Insurance coverage for Takeda Pharmaceuticals Korea's Zejula Cap (ingredient: niraparib tosylate monohydrate), a treatment for advanced ovarian cancer, will be expended, and the ceiling price of the drug will be reduced, starting on October 1st. Patients with advanced ovarian cancer who paid KRW 41 million in annual treatment costs per person are expected to pay KRW 2..05 million in annual treatment costs with the National Health Insurance coverage. On September 26th, the Ministry of Health and Welfare (MOHW) held the 19th Health Insurance Policy Review Committee meeting and decided on items related to National Health Insurance coverage of COVID-19 treatments and expanded National Health Insurance coverage for Zejula Cap. COVID-19 drugs will be covered by NHI…expanded reimbursement for Zejula The two COVID-19 treatments that the Korea Disease Control and Prevention Agency (KDCA) purchased and supplied will be covered by the National Health Insurance, starting in October. Those two are Paxlovid and Veklury. The ceiling price for Pfizer Korea's 'Paxlovid' is KRW 941,940 per 30 tablets in a package, and that for Gilead Sciences Korea's 'Veklury 100mg Powder For Concentrate For Solution For Infusion' is KRW 520,000 per vial. The related policy will be revised along with the reimbursement listing of these drugs. As an administrative action to prevent patient co-payment increases due to reimbursement, the co-payment will be maintained at the current cost of KRW 50,000. Starting next month, the reimbursement coverage for Zejula Cap, a treatment for advanced ovarian cancer, fallopian tube cancer, and primary peritoneal cancer, will be expanded, and its ceiling price will be reduced. For Zejula Cap, the usage criteria will be expanded, and it will be reimbursable for maintenance therapy of patients with advanced epithelial ovarian cancer, fallopian tube cancer, and peritoneal cancer who are responsive to the first-line platinum-based therapy. Previously, it was reimbursable only when the patients tested BRCA mutation-positive ovarian cancer from genetic screening. Starting next month, the drug will also be reimbursable in all homologous recombination deficiency-positive genetic mutations, including ovarian cancer-associated genomic instability identified from genetic testing. Patients with advanced ovarian cancer who paid KRW 41 million in annual treatment costs per person are expected to pay KRW 2..05 million in annual treatment costs with the National Health Insurance coverage. The pilot project for primary healthcare home visits will be improved Since December 2019, the government has been implementing a pilot project for primary healthcare home visits, a healthcare service in which private clinic doctors visit patient homes, to enhance healthcare access for patients who have difficulties visiting medical centers. Starting in November, the designation of medical centers for healthcare home visits will be expanded to include hospitals (local medical centers). Peviously, only private clinics and oriental medicine clinics were eligible for designation. Now, hospitals (local medical centers) can also participate following the policy revision. It will be expanded for patients with severe disease who require home medical care, including bedridden patients with Grade 1·2 long-term care and patients with severe disease using medical devices (oxygen therapy and a respirator). Co-patients for home visits will be reduced for these patients. In order to implement a reduction in out-of-pocket costs for eligible patients, a medical center's screening process will be developed. Additionally, a computer system for patients to claim the reduction of out-of-pocket costs is also in development. The implementation of the out-of-pocket cost reduction is scheduled to occur after November. In addition, to expand the primary healthcare home visit project, additional participating institutions will be invited in October. Extended NHI support for emergency care The policy for providing the National Health Insurance coverage for emergency care, which costs KRW 208.5 billion per month, will be extended to prevent a gap in healthcare for patients with severe and emergent symptoms amid the doctor's strike. The budget will be used to strengthen compensation for emergency departments and tertiary general hospitals so that they can focus on the treatment of emergency and patients with severe symptoms, and for the return of patients with mild symptoms to hospitals and clinics. Incentives will be increased to ensure the prompt transport of critical patients to hospitals. Additionally, incentives for medical care in emergency centers, including emergency care fees and CPR fees, will be enhanced. To facilitate prompt hospital responses to critical patient emergencies, the government will provide financial support for specialists treating hospitalized critical patients and incentives for critical patient hospitalization. The emergency medical care analysis comparing year-over-year from March to July 2024 shows that hospitals are maintaining critical patient care systems. However, the number of critical patients visiting the regional·local emergency medical centers has slightly decreased. The government will extend additional support to strengthen emergency care treatment capacity. The government will also extend raises for specialist medical fees, which were temporarily implemented during Chuseok to maintain the infrastructure of critical·emergency patient treatment at emergency medical centers. The raises for critical·emergency surgeries will also be extended. The raises in specialist medical fees will be 250% for regional·special emergency medical centers and 150% for local emergency medical centers. The raises for critical·emergency surgeries at regional·special·local emergency medical centers will be 200%. The MOHW stated, "We will provide a temporary insurance fee for emergency medical care to prevent a medical care gap for emergency and critical patients. We aim to promptly solve the current situation to ensure people do not encounter difficulties using medical centers."
- Company
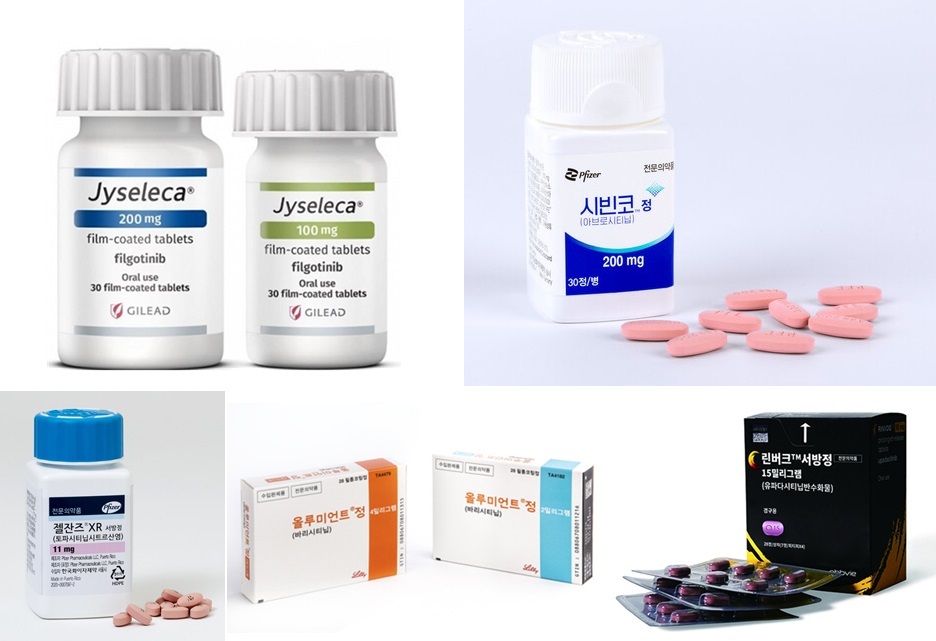
- Switching between JAKis for RA yet to be reimbursed
- by Eo, Yun-Ho Sep 27, 2024 05:52am
- 국내 처방되고 있는 JAK억제제들The plan to allow insurance reimbursement when switching between JAK inhibitors in rheumatoid arthritis, which was expected to take effect in October, has been postponed. According to Dailypharm coverage, the health authorities have recently put on hold the notification of the proposed revision that allows switching between JAK inhibitors in rheumatoid arthritis. The tentative timeline for the notification was set as the end of the year, but this is not confirmed yet. The delay was reportedly caused by problems with the final approval of the budget bill. Currently, 4 drugs are prescribed for rheumatoid arthritis in Korea, including Pfizer Korea’s Xeljanz (tofacitinib), Lilly's Olumiant (baricitinib), AbbVie's Rinvoq (upadacitinib), and Eisai Korea’s Jyseleca (filgotinib). Until now, the government has been adhering to the position that it is difficult to reimburse switching JAK inhibitors due to the lack of clinical evidence. However, after continuous statements submitted by the Korean College of Rheumatology and other organizations, as well as prescription experience on cross-dosing, the government reconsidered its position and came to a positive conclusion. The industry expected the change to be applied in October, but the notification of the amendment has been delayed. As a result, it remains to be seen if patients will be able to freely switch between JAK inhibitors before the end of the year. If reimbursement for switching between the drugs is allowed, such use is expected to further activate the JAK inhibitor market. According to drug market research firm UBIST, the outpatient prescription market for JAK inhibitors was KRW 27.5 billion in the first half of last year. This is a 54% increase in one year compared to the KRW 17.8 billion in the first half of last year. The market for JAK inhibitors has been expanding at a rapid pace. The market, which had been around KRW 12.5 billion in 2019, had expanded to KRW 18.7 billion in 2020, KRW 25.5 billion in 2021, KRW 33.5 billion in 2022, and KRW 40 billion last year. This year, the market reached KRW 27.5 billion in the first half of the year alone and is expected to exceed KRW 50 billion by the end of the year.
- Policy
- 2nd Approval-Evaluation-Negotiation pilot project imminent
- by Kim, Jin-Gu Sep 26, 2024 05:51am
- The initiation of the 2nd pilot project for the ‘approval-evaluation-negotiation linkage system,’ which was introduced to shorten the time from drug approval to reimbursement, is imminent. The Ministry of Health and Welfare has completed receiving drug submissions for the 2nd pilot project from pharmaceutical companies and is in the final review stage. The MOHW plans to implement the 2nd pilot project after reflecting on the improvements identified during the 1st pilot project. Eun-Joo Lee, an official from the Ministry of Health and Welfare’s Division of Pharmaceutical Benefits, announced so regarding the pilot project linking the approval-evaluation-negotiation system at the ‘Roundtable on Improving Access to Rare Disease Drugs' that was held at the National Assembly on the 24th. The government implemented the 1st pilot project for the approval-evaluation-negotiation linkage system last year. The project consists of simultaneous approval by the MFDS, reimbursement evaluation by the Health Insurance Review and Assessment Service, and drug price negotiations by the National Health Insurance Service. The project shortens the 300-day review period required for new drug access in Korea – which consists of an MFDS approval period of 120 days, 150 days of reimbursement evaluation by HIRA, and 60 days of drug price negotiations by the NHIS - and strengthens Korea’s access to new drugs. Two drugs - Qarziba Inj (dinutuximab, Recordati) and Bylvay (odevixibat, Ipsen) – were selected for the 1st pilot project. At the time, the government proposed the following criteria for selection: ▲a drug for cancer or rare diseases with a life expectancy of less than one year; ▲has a small number of patients; ▲lacks alternative drugs; ▲has a survival period over two years and demonstrated a superior treatment effect. Several companies submitted applications to participate in the pilot program, among which the government ultimately selected 2 drugs that met the selection criteria. Although the 1st pilot project has not been completed yet, the Ministry of Health and Welfare has been promoting to launch of the second pilot project. “We have completed receiving applications for the 2nd pilot project. We now need to select the drugs that meet the conditions of the 2nd pilot project,” said Lee. He added, “The number of drugs for the 2nd pilot project has not been decided upon yet. There are no criteria that require us to select only 2 drugs as in the 1st project. We will select drugs that meet the conditions.” The Ministry of Health and Welfare has proposed 4 criteria for selecting drugs for the 2nd pilot project: ▲ drugs that can apply for approval and decision by the end of June next year; ▲ drugs with sufficient effect for the treatment of life-threatening diseases (life expectancy of less than 1 year) or rare diseases; ▲ drugs that have no existing treatment or have shown clinically significant improvements in efficacy over existing treatments; and ▲ drugs that have been designated or are eligible for Global Innovative products on Fast Track (GIFT). To be selected as a drug for the 2nd pilot project, the drug needs to satisfy all the conditions above, and the company needs to submit the following data: ▲estimated financial expenditure per capita and total estimated financial expenditure; ▲results of overseas reimbursement evaluations; ▲the number of listed overseas countries; ▲the name of listed countries; and ▲name of the company. The MOHW plans to analyze the pros and cons of the 1st pilot project and apply them to the 2nd pilot project. In the 1stpilot project, the approval and reimbursement evaluation processes for Qarziba have been completed. The agenda had passed the Drug Reimbursement Evaluation Committee review and is currently being reviewed upon the company's reapplication. Compared to the existing system, the time from its approval to reimbursement evaluation has been significantly reduced. However, in the case of Bylvay, there are some delays due to difficulties in the evaluation process. The marketing authorization was completed on the 23rd of last month, but the reimbursement evaluation process was hampered due to the use of a difficult evaluation tool. However, the government is focusing on shortening the timeframe by conducting pre-negotiations at the same time. The HIRA explained that it will focus on how the companies manage uncertainties during the reimbursement evaluation process in the 2nd pilot project “In the future, uncertainty management will be important in the reimbursement evaluation process,” said Kook Hee Kim, Director of the Pharmaceutical Benefits Management Division at HIRA. ”Since the drugs are expected to be effective but not yet certain, the pharmaceutical companies should discuss how to address the uncertainties and prove their drug’s effect in the post-marketing process after the fact and share the risk with the government.” “The approval-evaluation-negotiation process is not a guarantee that the drug will be reimbursed. It means that the evaluation period is shortened,” explained Kim. ”The companies should submit data on how they will prove the uncertainties in the post-marketing process. Otherwise, the drug’s reimbursement may be delayed.”
- Company
- Minjung Jung to head Corporate Affairs at Sanofi KR, NZ, AU
- by Eo, Yun-Ho Sep 26, 2024 05:51am
- Sanofi’s Executive Director Minjung Jung (48) has been named head of Corporate Affairs for the Sanofi Group's 3 subsidiaries - Korea, New Zealand, and Australia. The appointment follows the appointment of Kyungeun Bae (53) as the General Manager of Pharma MCO South Korea and Australia/New Zealand & MCO Lead in the first half of last year, raising the stature of Sanofi Korea. With this promotion, Jung will now lead Corporate Affairs for the three countries, effective September 9th. The Corporate Affairs department is responsible for Public Affairs, Communications, CSR, Government, and Affairs. Jung is a seasoned public affairs professional who joined Sanofi in September last year. Since starting her career at MSD Korea in 2007, Jung has served various marketing roles at Boehringer Ingelheim Korea, Merck Korea, and Amgen Korea. Sanofi has been executing a Play To Win strategy, which focuses on building innovation platforms to develop first-in or best-in-class therapies and vaccines from 2020. The plan is to drive long-term growth through a virtuous cycle of efficiently redeploying resources to accelerate innovation and maximize R&D productivity. As a key part of this strategy, Sanofi has been operating its Korea and Australia & New Zealand subsidiaries as one integrated organization.
- Opinion
- [Reporter’s View] How to increase the new drug approval fee
- by Whang, byung-woo Sep 26, 2024 05:51am
- The Ministry of Food and Drug Safety (MFDS) decided to significantly raise its new drug approval fee to expand its capability for the prompt review of newly developed drugs. The fee for new drug approval, which was previously KRW 8.83 million, was increased 48 times to KRW 410 million based on the benefit principle. Despite the dramatic increase, there has been little backlash as Korea’s drug approval fee has been relatively low compared to overseas. The MFDS aims to raise the new drug approval fee from next year after a 60-day administrative notice period. As the pharmaceutical industry has been actively supporting the realization of the fee, there have been more expectations than concerns. Rather, the industry's concern is whether the fee hike will have an immediate effect. The MFDS plans to use the increased fees to recruit specialized review personnel and expand the proportion of high-competency reviewers, such as doctors, pharmacists, and those with at least 3 years of postdoctoral experience, from the current 31% to 70%. However, whether the government can secure the required specialized personnel remains in question. While the MFDS emphasizes the importance of securing human resources by allocating half of the fee hike to labor costs, it is unclear at what point it will be able to recruit the desired amount of human resources. This is also a concern for pharmaceutical companies. While there is no doubt that the fee hike will speed up approvals, there is also concern that pharma companies will not immediately see the benefits of their inputs. As a result, some pharmaceutical companies are considering accelerating their submission plans and applying for approval before the fee hike. For example, there are concerns about whether the review process for new drug approvals will change dramatically before and after the fee increase, or whether the new system will take time to settle in. In the end, the pharmaceutical industry needs the MFDS to preemptively establish a system so that the increased fees can be utilized as intended. Apart from the consensus on the KRW 400 million increase in review fees, the system should not have a backward-looking investment structure that raises fees and then uses them to increase the number of reviewers. With the expected improvement in the system is expected, attention is likely to be focused on the first case of their application. In the bigger picture, the MFDS is hoping that the fee hike will also prevent the poke-and-see applications without sufficiently demonstrating safety and efficacy, in addition to shortening the review period. In other words, it could act as a hurdle to prevent companies from using Korea’s regulatory authorities as sort of a consulting window. However, the pharmaceutical industry is also hoping that the MFDS’s review will also include consultation at the approval stage, as the authorities are requesting new drug fees at the level of FDA and EMA. The increase in drug approval fees is expected to bring about many changes in the domestic regulatory environment. As the saying goes, 'the beginning is half the battle', so we expect thorough preparation and changes to be made to meet the purpose of the MFDS’s new drug approval innovation plan.
- Policy
- Higher dosage Eylea expected to strike back at biosimilars
- by Lee, Tak-Sun Sep 26, 2024 05:51am
- The competition in the market for the macular degeneration drug Eylea has recently intensified with Samsung Bioepis and Celltrion, which have recently launched biosimilars. The original developer is set to strike back against the competitors. Bayer will release Eylea Inj 8 mg (aflibercept), with an extended treatment interval, into the market next month at a relatively cheaper price. According to the industry on September 25th, Eylea Inj 8 mg will be listed for reimbursement next month at a price lower than the evaluation price, KRW 795,000. Eylea Inj 8 mg provides a long-lasting, effective dosage within the eye at a four times higher dosage than the previous 2 mg. It extends the treatment interval while reducing the number of injections. Eylea Inj 8 mg is injected once monthly for an initial 3 months. Then, based on visual and anatomical test outcomes, a treatment interval can be extened up to 16 weeks at the physician's judgement. After the initial extension of the treatment interval, it can be extended up to 20 weeks with the Treat-and-Extend(T&E) therapy. This approach allows flexibility in adjusting the treatment interval depending on the patient's condition while maintaining stable vision and anatomical test outcomes. The original and biosimilars currently sold in the market are at a 2 mg dosage. For neovascular age-related macular degeneration (nAMD) treatment, the drugs are injected once monthly for an initial 3 months, followed by a once every 2 months treatment regimen. Using the new 8 mg will extend the treatment interval more than twice, from once every 2 months to once every 4 months. The PULSAR study secured non-inferiority results of the new product by showing that patients treated with Eylea 8 mg at a 12-week interval had the best corrected visual acuity (BCVA) of 6.7 letters on average from the baseline at 48 weeks, those at a 16-week interval had BCVA of 6.2 letters, and those at an 8-week interval had the BCVA of 7.6 letters. However, the drug price is not more than double the original price. Due to the biosimilar release, the current ceiling price for Eylea Inj·Prefilled Syringe is KRW 496,118, a 30% reduction from the highest price. The newly listing Eylea Inj 8 mg costs KRW 795,000. Despite extending the treatment interval more than twice, the price has not doubled because Bayer has set the price lower than the evaluated price. However, the price is double that of biosimilars. Samsung Bioepis' Afilivu Inj 40 mg/ml costs KRW 350,000, and Celltrion's Eydenzelt Inj costs KRW 330,000. Although the price of biosimilars is low, newly diagnosed patients are likely to choose the high-dose original drugs, which require fewer injections due to the extended treatment interval. Hence, Eylea Inj 8 mg will likely be a direct competitor to biosimilars. Eylea is a blockbuster product, recording KRW 96.8 billion in sales last year, based on IQVIA. Due to its marketability, Samsung Bioepis and Celltrion, which have released their products in May and September, respectively, are trying to seize the market by partnering with pharmacists who are specialists in ophthalmology. Industry personnel said, "Patients who are benefiting from the existing Eylea 2 mg are less likely to switch products. However, new patients are likely to choose from biosimilars, with lower cost, and high-dosage original drug, with fewer injections," adding, "An intense competition among overseas original developer and Korean biosimilar developers is expected."
- Policy
- Potential expanded indication for obestiy drug 'Wegovy'
- by Lee, Hye-Kyung Sep 26, 2024 05:51am
- Product photo of Wegovy. A clinical trial for Novo Nordisk Pharma's obesity drug, 'Wegovy (semaglutide),' will be conducted in South Korea for expanded indication. On September 20th, the Ministry of Food and Drug Safety (MFDS) has approved the 'Phase 2 clinical trial to evaluate the efficacy, safety, and dosage testing of NNC0519-0130, subcutaneously administered once-weekly, in comparison to Ozempic (semagluide) 1.0 mg and placebo in overweight or obese patients who have or do not have type 2 diabetes.' The clinical trial will involve 456 individuals worldwide from December 2024 to September 2026. In South Korea, the study will enroll 32 patients and will be conducted at Pusan National University Hospital, Ilsan Hospital, Hallym University Medical Center, Seoul St. Mary's Hospital, Hanyang University Guri Hospital, and Korea University Guro Hospital. Wegovy received domestic marketing authorization in April with indications to ▲aid weight-loss overweight patients who have a Body Mass Index (hereafter referred to as BMI) of 30kg/m2 or higher or those who are overweight with early BMI of 27kg/m2 or higher and below 30kg/m2 and having one or more weight-related accompanying diseases (abnormal glycemia, type 2 diabetes, high blood pressure, dyslipidemia, obstructive sleep apnea, or cardiovascular diseases) ▲reduce risks of major cardiovascular events in overweight or obese patients with early BMI of 27 kg/m² or higher with cardiovascular diseases. Additionally, the company will conduct the Phase 2 clinical trial to add indication to treat chronic kidney disease. Based on Phase 3 SELECT study results, presented by Dr. Katherine Turtle, a professor at the University of Washington School of Medicine, at a recent European Renal Association meeting, Wegovy can reduce the risk of kidney failure and death in patients with chronic kidney disease because of type 2 diabetes. The clinical trial involved 3533 patients with type 2 diabetes and chronic kidney disease. Half of the participants received weekly semaglutide injections for 3 years, and the rest received weekly placebo. Patients treated with semaglutide had a lower possibility of worsening symptoms that require dialysis or kidney transplant by 24% and a lower possibility of death due to major cardiovascular disease, including heart attack, than those treated with placebo by 29%. Patients treated with semaglutide had a 20% lower possibility of death of all causes during the study period. However, investigators studying semaglutide have not found an exact mechanism by which this drug helps the kidney, and the study was limited in that around two-thirds of the participants were white males. Meanwhile, Wegovy's active ingredient, semaglutide, is a 'Glucagon-like protein (GLP)-1' agonist. GLP-1 receptors lower blood pressure and regulate appetites. The basis for Wegovy's approval in South Korea was the STEP study, in which patients treated with Wegovy (1306 patients) had a 14.9% weight loss across 68 weeks compared to the baseline, showing a significant difference from a 2.4% of the placebo group (655 patients). According to Novo Nordisk Pharma's business performance reporting for the first half of 2024, Wegovy generated US$3.07 billion, up 74% year-over-year.