- LOGIN
- MemberShip
- 2026-04-04 15:20:52
- Policy
- Celltrion drops export permit for FDA-Cleared HIV drug
- by Lee, Tak-Sun Jan 23, 2026 08:39am
- Celltrion has withdrawn the Korean export authorization for an HIV-1 infection treatment drug approved by the U.S. Food and Drug Administration (FDA).The company had pursued overseas HIV treatment markets, including the U.S., through the export authorization, but the move is being interpreted as a change in business strategy. Going forward, Celltrion is expected to focus more heavily on its biosimilar business.According to the Ministry of Food and Drug Safety, Celltrion Pharm withdrew the export license for ‘Temixys Tab 300/300mg (lamivudine/tenofovir disoproxil fumarate)’ as of the 7th. It also withdrew the export approval for the triple-combination HIV drug ‘Telumio Tab (tenofovir disoproxil fumarate, lamivudine, dolutegravir sodium)’.Temixys is a combination drug that combines the active ingredients of GSK's existing original antiviral drug, Zeffix (lamivudine), and Gilead's antiviral drug, ‘Viread (tenofovir)’. It is a product developed by Celltrion for use in combination with other antiretroviral agents to treat HIV-1 infection in adults and children weighing at least 35 kg.Celltrion announced that it received FDA approval for the sale of Temixys on November 16, 2018. This signaled competition with Gilead's ‘Truvada’ in the U.S. HIV treatment market.Prior to FDA approval, Celltrion had obtained export drug authorization from the Ministry of Food and Drug Safety on October 25 of that year, completing preparations for overseas sales.At the time, Celltrion stated it would supply high-quality treatment at a reasonable price compared to the original drug to HIV patients in the U.S. who were unable to access adequate medication due to high drug prices and insurance structures.The company also expressed plans to enter the international procurement market by securing supplier qualification from major global HIV drug procurement organizations, including the World Health Organization (WHO), the Global Fund, USAID, and the United Nations agency UNDP.To this end, the Celltrion Group explained that it had established a new chemical development team within Celltrion and launched a global chemical project.However, the withdrawal of this export license for the AIDS treatment suggests a shift in the company’s business strategy, 7 years after FDA approval. Domestic production of the AIDS treatment will now be discontinued following the authorization withdrawal. Celltrion is expected to focus more intensely on its biosimilar business going forward.
- Company
- Pharma in Hyangnam "Unstable employment cannot yield good medicine production"
- by Chon, Seung-Hyun Jan 23, 2026 08:39am
- Landscape of Hyangnam Pharmaceutical ComplexThe pharmaceutical industry gathered at the Hyangnam Pharmaceutical Complex to appeal for a halt to the government's drug price reduction plans. Workers at the complex, where many small and mid-sized pharmaceutical companies are located, expressed fears of job instability, and the possibility of future labor struggles was raised.The "Emergency Response Committee for Drug Price System Reform for the Development of the Pharmaceutical and Biotech Industry" (the Committee), composed of major industry organizations, held a labor-management meeting on the 22nd at the Hyangnam Pharmaceutical Complex in Hwaseong, Gyeonggi Province. The Committee requested a reconsideration of the government's reform. The committee is consisted of the Korea Pharmaceutical and Bio-Pharma Manufacturers Association (KPBMA), the Korea Biomedicine Industry Association, the Korea Pharmaceutical Traders Association, the Korea Drug Research Association, and the Korea Pharmaceutical Industry Cooperative.The event was organized for the Committee and the labor-management of the Hyangnam complex to review the risks and impact that the reform plan, which includes large-scale price cuts, would have on the industry and production sites.The event's participants warned, "The large-scale drug price cuts pushed by the government are a declaration of abandonment of the future of the domestic pharmaceutical industry, run counter to the demand of these days for job stability, and collapse the national health safety net."In November last year, the MOHW reported to the Health Insurance Policy Review Committee its plan to lower the price calculation rate for generics and patent-expired drugs from 53.55% to around 40%. The reform is scheduled to be finalized by the Health Insurance Policy Review Committee in February and implemented in July. A month after a press conference held at the KPBMA in Seocho-gu on the 22nd of last month, the pharmaceutical industry once again publicly stated dissatisfaction against the government.The "Emergency Response Committee for Drug Price System Reform for the Development of the Pharmaceutical and Biotech Industry" (the Committee), composed of major industry organizations, held a labor-management meeting on the 22nd at the Hyangnam Pharmaceutical Complex in Hwaseong, Gyeonggi Province. Participants stated, "If the government's reform is implemented as is, it will result in the virtual collapse of the domestic pharmaceutical industry and lead to fatal consequences that jeopardize the health and lives of the public." Hyangnam Pharmaceutical Complex is Korea's largest pharmaceutical production hub, housing 39 business sites from 36 companies.Attendees unanimously agreed that rapid price cuts could deal a severe blow to the entire industry, including job instability, a contraction in R&D investment, and a weakened production base.Participants stated, "If rapid and unprecedented price cuts are implemented, the damage will be concentrated on domestic pharmaceutical companies, including those in Hyangnam, making a loss of up to KRW 3.6 trillion." The industry fears that deteriorating profitability will hinder investment in facilities for quality innovation, infrastructure improvements, and R&D.Oh Sang-jun, Chairman of the Southern Gyeonggi branch of the Chemical Workers' Union under the Federation of Korean Trade Unions (FKTU), voiced his opinion, saying, "Workers may fear job instability due to the government's price cuts. If employment is unstable, good medicine cannot be produced. The government should stop pushing unilaterally and instead consult with pharmaceutical workers to implement an effective drug pricing policy. If necessary, we will resort to struggle."Labor and management representatives from the five organizations in the Committee predicted that more than 10% of the industry's 120,000 workers would inevitably face unemployment. They also pointed out that a contraction in the production of low-profit essential medicines and domestic ethical drugs would ultimately increase reliance on expensive imported drugs, causing serious problems for public health.The labor-management representatives emphasized, "Stop the unilateral push for drug price cuts and guarantee job stability in the domestic pharmaceutical industry," and added, "Officials in charge of healthcare safety should actively foster the industry." Officials from companies in the Hyangnam complex also repeatedly highlighted the side effects.Wonsuk Lee, CEO of Daehan Nupharm, urged, "The reform plan is becoming a profitability shock to small and mid-sized domestic pharmaceutical companies, highly dependent on generics. Small businesses will be unable to handle even the fixed costs for facility investment and quality control. Before pushing an unreasonable plan that ignores the industry's reality, please prepare a reasonable alternative through communication."Lee Deok-hee, Chairman of the Il-dong Pharmaceutical labor union, said, "We have had the unfortunate experience of conducting restructuring due to liquidity limits while continuously making bold investments of 20% of sales to grow future competitiveness," and added, "Price cuts will lead to permanent workers becoming temporary workers and indirect employment leading to dismissals, which could greatly shake the job stability and the very existence of the pharmaceutical industry."On this day, the Medicine and Cosmetics Division of the National Federation of Chemical Workers' Unions under the FKTU issued a statement calling for a "full reconsideration of the drug price system reform and the protection of pharmaceutical jobs."The Medicine and Cosmetics Division urged: ▲reconsideration of the reform, ▲the establishment of a social discussion body involving pharmaceutical workers and unions, and ▲the preparation of measures to protect jobs and ensure employment stability.Lee Dong-in, Executive Secretary of the division, suggested the possibility of a struggle, stating, "We will respond strongly against job instability and restructuring caused by the reform," and added, "We will inform the public of the voices of pharmaceutical workers through various means and continuously raise issues regarding the reform plan."Yunhong Noh, President of the KPBMA, argued, "If the reform is forced through in its original form, the industrial foundation will collapse, and the production of essential medicines will contract. Hyangnam, which is home to small- and mid-sized companies, could face job instability and economic contraction due to changes in the business environment," and "A policy that ignores the voices of the industrial field can never succeed."Cho Yong-jun, Chairman of the Korea Pharmaceutical Industry Cooperative, appealed, "The generic drug price reform plan could pose serious business risks to small and mid-sized pharmaceutical companies," and added, "Rather than a unilateral cut, a phased approach is needed after analyzing the practical impact on employment and investment. Rapid change destroys even the best policy's ecosystem. Companies should be allowed enough time to improve their system."
- Policy
- Ozempic, Rezurock, HyalFlex covered from next month
- by Lee, Jeong-Hwan Jan 23, 2026 08:39am
- Novo Nordisk’s type 2 diabetes treatment Ozempic (semaglutide) and Sanofi’s graft-versus-host disease (GVHD) treatment Rezurock (belumosudil) will be covered by Korea’s National Health Insurance starting on the 1st of next month.In addition, new reimbursement standards will be set for Jeil Pharmaceutical’s antibiotic Fetroja (cefiderocol) and Shinpoong Pharmaceutical’s osteoarthritis treatment HyalFlex (hexamethylenediamine cross-linked sodium hyaluronate).On the 22nd, the Ministry of Health and Welfare announced an administrative notice for a partial amendment to the “Detailed Standards and Methods for the Application of Medical Care Benefits (Pharmaceuticals),” which includes the establishment of reimbursement criteria for Ozempic, Rezurock, HyalFlex, and Fetroja.The Ministry plans to gather opinions until the 26th and then submit the agenda for deliberation at the Health Insurance Policy Deliberation Committee meeting on the 29th.For Ozempic Prefilled Pen, new coverage criteria will be established for its use as an adjunct to diet and exercise therapy in combination with oral agents or insulin in adult patients with type 2 diabetes that is not adequately controlled.Coverage for its use as part of the oral combination therapy applies to patients with a glycated hemoglobin (HbA1c) level of 7% or higher despite 2-4 months of combined metformin and sulfonylurea therapy, provided their body mass index (BMI) is 25 kg/m² or higher, or they are unable to use insulin. These patients are allowed reimbursed use of a triple therapy that includes Ozempic.However, if blood glucose is significantly improved with triple therapy, dual therapy with only metformin may also be reimbursed.Its use in combination with insulin (+metformin) is covered when HbA1c remains 7% or higher despite 2-4 months of basal insulin (insulin alone or with metformin) or when HbA1c remains 7% or higher despite combination therapy with Ozempic and metformin (+sulfonylurea).However, objective documentation (treatment history, HbA1c, BMI, etc.) must be submitted at initiation. HbA1c and BMI must be monitored every three months.The coverage period per prescription is up to 4 weeks for the initial 3 months when dosage adjustment is necessary per the approved indications, then up to 3 months thereafter.Rezurock is reimbursed for adults and pediatric patients aged 12 years and older who have failed at least two prior systemic therapies (including ruxolitinib).If ruxolitinib cannot be used due to adverse reactions or contraindications, coverage applies after failure of two or more alternative systemic therapies.If disease progression is absent at the 6-month and 9-month evaluations, and a response is confirmed at the 12-month evaluation, an additional 3 months of treatment is approved with reimbursement.Thereafter, continuous administration is approved upon confirmation of response at each 3-month evaluation. Re-administration is also reimbursed for patients who discontinued due to improvement.However, treatment must be discontinued if GVHD progresses (symptoms worsen or new symptoms appear), unacceptable adverse reactions that render use of Rezurock impossible occur, or a new systemic therapy is initiated after Rezurock.In addition, objective data (such as medical records and blood test results) regarding the target of administration for the initial dose of Rezurock, as well as response evaluations for continued administration or discontinuation, must be submitted.In the case of HyalFlex, coverage applies to patients with knee osteoarthritis of radiographic severity Kellgren-Lawrence grade I–III (mild to moderate).In the case of Fetroja, coverage is applied for complex urinary tract infections and hospital-acquired pneumonia when treatment with carbapenem antibiotics has failed, or when multidrug-resistant Pseudomonas aeruginosa, carbapenem-resistant Enterobacteriaceae, or carbapenem-resistant Acinetobacter species are confirmed. And a physician’s treatment report must be submitted.For DongKwang Pharm’s Sulbacin Inj and other sulbactam/ampicillin combinations, coverage is approved beyond the approved indications when infection with carbapenem-resistant Acinetobacter baumannii is confirmed. The maximum daily dose is 27g, and the treatment period is within 14 days. Coverage may be approved for periods exceeding 14 days upon reference to the physician’s treatment report.In addition, Hanwha Pharmaceutical's Hepa-Merz Inj and other L-aspartic acid-L-ornithine preparations are covered when symptoms of latent or overt hepatic encephalopathy are present.The general principles for calcium and vitamin D-containing oral combination preparations now include hypoparathyroidism due to procedures such as total thyroidectomy.
- Policy
- Upcoming drug price reduction
- by Lee, Jeong-Hwan Jan 23, 2026 08:39am
- Major domestic pharmaceutical companies and mid-sized firms are repeatedly requesting modifications to the drug price reform plan, which was announced by the Ministry of Health and Welfare (MOHW) for implementation this year. The requests include a delay in its implementation and adjustments to the price reduction rates.The MOHW has yet to provide a clear response regarding the industry’s requests for a delay, changes to the reduction rates, or revisions to the preferential pricing details. Instead, the MOHW appears to be maintaining its "original plan to finalize the decision at the Health Insurance Policy Review Committee within the first quarter."In response, members of the National Assembly's Health Insurance Policy Review Committee, from the opposition party, plan to mediate between the pharmaceutical industry and the MOHW by hosting policy forums.On the 22nd, Rep. Baek Jong-heon of the People Power Party, alongside Reps. Han Ji-ah and Ahn Sang-hoon, will co-host a policy forum on drug price system reform on the 26th,. The forum will be organized by the Korea Pharmaceutical and Bio-Pharma Manufacturers Association (KPBMA).During the forum, Attorney Park Kwan-woo from Kim & Chang will analyze the impact of the MOHW's drug price reform on public health, and Attorney Kim Hyun-wook from Shin & Kim LLC will present on sustainable measures to improve the drug price system.In the subsequent panel discussion, representatives from large domestic pharmaceutical companies, as well as the Korea Pharmaceutical Industry Cooperative, representing mid-sized and small firms, will participate to demand a delay in implementation, adjustments to the reduction rates, and revisions to the MOHW's preferential pricing plan.Specifically, the industry representatives, including Yoon Jae-Chun, Vice Chairman of Daewoong Pharmaceutical; Young-Joo Kim, CEO of Chong Kun Dang Pharm; and Cho Yong-jun, Chairman of the Korea Pharmaceutical Industry Cooperative, will be attending. From academia and patient groups, Professor Hye-Young Kwon of the Department of Health and Medical Administration at Mokwon University and Yoon Gu-hyun, President of the Korean Association for the Study of Liver, will attend. Yeon-sook Kim, Head of the Pharmaceutical Management Division at the MOHW, will represent the government.Attention is focused on whether this forum, which aims to seek a balance between the sustainability of health insurance and the development of the pharmaceutical and biotech industries, can narrow the gap in perspectives between the industry and the government.Multiple industry sources are appealing to the MOHW to accept the industry's proposed modifications proactively. The top priority for the pharmaceutical industry is delaying the committee's approval of the reform plan, currently scheduled for next month (February).On November 28 last year, the MOHW publicly announced the reform plan, which centers on cutting generic drug prices, and reported the implementation schedule to the committee.Several domestic pharmaceutical companies maintain that the implementation plan is too rushed, failing to ensure the predictability necessary for business management.The pharmaceutical industry is also demanding that the price-reduction rates for already-listed drugs (generics) be minimized. While the MOHW plans to lower the current generic price calculation rate from 53.55% to the 40% range, the industry argues this cut is excessive and insists it should be set at 48% to 50%.Additionally, they have requested that the implementation timing for generics listed since the 2012 price cuts be pushed back sufficiently to ensure predictability, and that the details of the preferential pricing plan be revised through consultation with the industry.An official from a top-tier domestic pharmaceutical company said,"It is frustrating that specific consultations or coordination of opinions between the MOHW and the industry regarding the reform plan have not yet taken place," adding, "We have requested several times that the short time between the announcement and the implementation date undermines predictability. A delay in the committee's approval is necessary."Another official from a top-tier company added, "Although several forums regarding the drug price reform have been held in and around the National Assembly, the atmosphere remains unclear as to whether the government will modify its plan or accept the industry's demands. If the drug price reform aims to foster the domestic pharmaceutical industry, the MOHW needs to commit practical and proactive consultations with the sector."
- Company
- 'Drug pricing reform will improve patient access'
- by Son, Hyung Min Jan 22, 2026 08:22am
- As global pharmaceutical R&D trends rapidly shift toward high-priced anticancer and rare disease therapies, attention is focused on how the government's proposed drug pricing system reform will impact domestic patient access and the competitiveness of the pharmaceutical industry.On the 21st, Dailypharm held a Future Forum at the Catholic University of Korea's Songeui Campus Institute of Biomedical Industry. At the forum, Jaeho Jung, Head of Value and Access at Novartis, presented the perspective of multinational pharmaceutical companies on the proposed drug pricing reform.Jung said, “The reform plan is in line with global R&D trends. If appropriate value-based compensation and faster reimbursement listing are achieved, it will send a positive signal not only for patient access but also for Korea’s R&D ecosystem.”On the 21st, Dailypharm held its 55th Future Forum at the Catholic University of Korea's Songeui Campus Institute of Biomedical Industry.Development of high-cost anticancer drugs and rare disease treatments active... “Current drug pricing and reimbursement systems struggle to accommodate innovation.”Jung first highlighted the scale of clinical development in oncology and rare disease therapies. Currently, oncology accounts for 41% of all global clinical trials, of which 35% involve innovative drugs.The rare disease treatment market is expected to reach KRW 542 trillion by 2023, while rare cancer trials account for 74% of all ongoing rare disease studies.Despite this projected expansion of the high-cost new drug market, securing insurance coverage, which is a core value for patient access in Korea, remains inadequate. Among 460 new drugs approved in major countries from 2012 to 2021, the coverage rate averaged 28% across G20 nations and 29% in OECD countries, while Korea lagged at just 17%.Jung said, “Korea is facing a triple burden of ensuring universal welfare with limited resources, expanding access to new drugs, and guaranteeing patients’ right to treatment. The government has not been idle. The system has been continuously revised since 2007, but it has now reached its limits.”Examining the time required for reimbursement by treatment type, rare disease treatments took 23.6 months, anticancer drugs 31.6 months, and general new drugs 18.3 months. Even after new drug approval, a lengthy period remains before patients really gain reimbursement.At the same time, global R&D trends have driven the rise of antibody-drug conjugates (ADCs), bispecific antibodies, and biologics, many of which now hold multiple indications. Some immuno-oncology drugs have more than 20 approved indications.In response, the government has proposed reforms aimed at improving access to new drugs. These include an indication-based pricing, strengthening value-based drug reimbursement, and shortening the reimbursement listing period for rare diseases. In particular, the reimbursement listing period for rare disease therapies is expected to be reduced from an average of 240 days to within 100 days.Jung assessed, “With the proportion of high-cost new drugs rapidly increasing, it is difficult to guarantee patient access solely with limited national health insurance funds. As more drugs secure multiple indications, an indication-based pricing system is in line with global standards”He added, "The current system focuses on increasing reimbursement rates within the existing framework; it remains finance-centered from a patient access perspective. Although the government’s approach is understandable given budget constraints, the system must be operated with the patients in mind.”“Reform will create synergy, not suppress R&D”Jaeho Jung, Head of Value and Access at Novartis KoreaHe also expressed the view that the drug pricing reform could improve the domestic investment environment.Korean pharmaceutical companies, whose portfolios are largely centered on generics, have repeatedly voiced concerns that the proposed drug pricing reform could significantly stifle R&D.However, Jung emphasized the potential advantage that improved access to new drugs could expand investment by global pharmaceutical companies.He explained that currently, only two global pharmaceutical companies have production facilities in Korea, and attracting global-level R&D centers remains challenging.Jung stated, “Even when reviewing potential sites for our flagship one-shot treatment Kymriah, we received feedback that Korea was not suitable. If the system becomes more predictable and the value of R&D is properly recognized, direct investment and partnerships with Korean companies will expand.”He added, “If drug pricing improves in terms of value recognition and patient access, direct investment by global pharmaceutical companies will increase, as well as partnerships with Korean companies.”“Looking at Boston or New Jersey, which have rapidly emerged as global bioclusters, their R&D centers are well-established. This is because they are clustered together in terms of the collaborative R&D value. Looking at the domestic R&D environment, it feels like everyone is working hard, but individually. If the drug pricing reform system is well-established, I believe synergies will grow further, including joint R&D centers between global and domestic companies.”Furthermore, Jung pointed out that discussions on reimbursing high-value new drugs like ADCs and one-shot gene therapies have repeatedly faced difficulties within the existing framework. “The reform plan has a high potential to contribute to improving patient access, aligning with future global R&D trends.”Jung stated, “Operational execution is as crucial as the system's design. I hope a patient-centered system can take root through continuous dialogue between the government and industry.”
- Policy
- 'Key is in compensating innovation that exceeds price cuts'
- by Lee, Jeong-Hwan Jan 22, 2026 08:22am
- Director Yeon-sook Kim explains the drug pricing system reform plan.“While the media has largely focused on how the upcoming drug pricing system reform plan involves adjustments and reductions in drug prices, the true core of the policy is a fundamental strengthening of innovation-based compensation for pharmaceutical R&D beyond mere adjustments Whether it's a new drug or a generic, if it demonstrates innovation, we will increase drug price compensation and extend the preferential period from the current one year to three years plus alpha, creating a virtuous cycle of innovation in the pharmaceutical industry.”The Ministry of Health and Welfare reiterated its stance that the core keywords of the drug pricing system reform plan announced for implementation this year are ‘innovative new drugs’ and ‘resolving supply instability,’ and that it will enhance the tangibility of preferential drug pricing incentives for pharmaceutical companies that achieve these goals.This involves establishing a drug pricing compensation structure proportional to the level of investment in new drug R&D and extending the preferential period from the current one year to ‘three years or more.’Regarding resolving supply instability, the government announced plans to guarantee preferential treatment for up to 10 years for drugs using self-manufactured APIs and for essential medicines made with domestic APIs.The Ministry's policy is to overhaul the post-listing drug price control system to enhance predictability for pharmaceutical companies while reducing the administrative burden on the government.Kim outlined the reform and its operational roadmap on the 21st at the 55th Dailypharm Future Forum held at the Catholic University of Korea’s Songeui Campus Institute of Biomedical Industry.“The current pricing system has limitations in supporting innovation and essential drug stability”The Ministry of Health and Welfare maintains that the current drug pricing system has clear limitations in reliably ensuring the creation of innovative new drugs and stable patient access to essential medicines.It was further diagnosed that, compared to other countries, South Korea's generic drug prices are relatively high, leading to stagnation in innovation through new drug R&D.The ministry believes that many pharmaceutical companies remain entrenched in business models centered on high generic drug prices, neglecting new drug R&D.At the same time, population aging and rising pharmaceutical expenditures have significantly increased drug spending, making it increasingly difficult to strike a balance between fostering an innovation-based pharmaceutical industry ecosystem and ensuring the sustainability of the National Health Insurance system.“Drug price reform to foster a new drug ecosystem and ensure a stable supply of essential medicines”To strengthen the new drug development ecosystem, the ministry plans to shorten the reimbursement listing period for rare disease treatments to within 100 days and to advance cost-effectiveness evaluations for treatments targeting severe and intractable diseases.To facilitate the early domestic introduction of innovative medicines, the scope of the flexible drug pricing contract system should be expanded, and compensation should be provided proportional to the level of innovation creation efforts, such as new drug R&D.This includes strengthening R&D-linked pricing incentives and ensuring stable preferential periods.For essential medicines, the ministry will overhaul the exit-prevention drug system across the entire lifecycle, strengthen the effectiveness of preferential treatment for essential drugs, and reinforce coordination between relevant policies.Through a public–private consultative body, supply stability medicines will be monitored throughout their lifecycle and addressed proactively.The government will also strengthen monitoring of pharmaceutical supply and distribution and implement tailored measures based on the causes of shortages.In particular, for medicines with unstable supply, the government plans to support substitution prescribing and dispensing at the national level.“Generic pricing rate to be adjusted to the 40% range… stepwise cuts to apply from the 11th product”The most controversial part of the reform among domestic pharmaceutical companies is the rationalization of price management.The ministry plans to revise the pricing structure based on international benchmarks, with the key change being a reduction of the generic pricing rate from the current 54.55% to the 40% range.The stepwise price reduction system will also be strengthened. Currently, stepwise cuts apply from the 21st product of the same ingredient, with the price set at 85% of the lowest price.Under the reform, price cuts will apply from the 11th product, and the reduction rate will be set at an additional 5 percentage points below the first generic price.The Ministry of Health and Welfare anticipates that this reform will significantly increase the perceived benefits for pharmaceutical companies by applying price premiums primarily to those contributing to new drug innovation and supply stability.For price reductions of already-listed generics, items maintaining a price level of 53.55% since the 2012 blanket price reduction adjustment will be sequentially reduced over three years.The post-listing management system will standardize the timing for adjusting price reductions linked to expanded usage and price-volume linkage agreements. Actual transaction price surveys will be restructured to a market competition-linked model. The reimbursement adequacy reassessments will be restructured around clinical evidence.Kim emphasized, “Korea is at a critical moment, both a crisis and an opportunity, to build an innovative new drug ecosystem. We will raise pricing rewards for innovation and significantly extend preferential periods to send a clear signal toward innovation.”She added, “Even if a product is not a new drug, it will receive preferential pricing if it contributes to a stable supply of essential medicines. For exit-prevention drugs, we will collaborate with organizations like the Korea Pharmaceutical and Bio-Pharma Manufacturers Association and the Health Insurance Review and Assessment Service to identify tasks for improving drug pricing levels or systems after conducting research studies. We will ensure that the number of generic items remains at an appropriate level, avoiding excessive proliferation.”“When too many products of the same ingredient are listed at once, prices will be adjusted according to stepwise criteria after an appropriate period. For already-listed products, prices that have remained high since 2012 will be reduced gradually and sequentially over three years.”
- Policy
- Gov't addresses unstable supply of essential drug·API
- by Lee, Jeong-Hwan Jan 22, 2026 08:22am
- To strengthen response to drug supply instability this year, the government is expanding support for pharmaceutical companies producing items in short supply, improving self-sufficiency in active pharmaceutical ingredients (APIs), and enhancing the manufacturing capabilities for biopharmaceutical raw materials and components.On the 20th, Yim Kang-seop, Head of the Division of Health Industry at the Ministry of Health and Welfare (MOHW), met with the Korea Special Press Association to explain measures to address the supply chain crisis facing the pharmaceutical and biotech industries.To address structural limitations in the drug supply system, the MOHW has secured a total budget of KRW 15.6 billion for this year and is pursuing five projects.The projects include support for production facilities and equipment for companies supplying drugs with unstable supply, support for stockpiling core medicines, support for diversifying API procurement, user testing support for bio-based raw materials and components, and manufacturing support for domestic raw materials and components.First, the MOHW will increase the number of pharmaceutical companies receiving budget support for producing drugs with unstable supply from 1 last year to 4 this year.This project, launched for the first time last year, currently supports 'Boryung Questran Powder For Suspension (Cholestyramine resin),' a bile acid sequestrant-based hyperlipidemia treatment, and the only drug safe for use by pregnant women and children.Yim plans to reorganize the program to allow subsidy support for up to two years, taking into account the actual time required for pharmaceutical companies' demand and equipment setup, while also increasing the number of target items.To produce API domestically, new projects starting this year include support for diversifying raw material procurement, which helps consortia between raw material suppliers and finished product manufacturers, and a project to cover stockpiling costs to ensure a stable supply of medicines even during crises.In collaboration with the Korea Health Industry Development Institute (KHIDI), the MOHW will also launch initiatives for user testing and manufacturing support to promote the use of domestically produced biopharmaceutical raw materials and components.Yim explained, "Korea's self-sufficiency rate for API is currently low, in the low 20% range. He noted that dependence on specific countries, such as China and India, is high, and that supply chain risks are growing as countries shift toward protectionist trade policies," adding, "To resolve these structural issues, the ministry is launching new projects this year to directly support supply chain stability, including the localization of raw materials and components, stockpiling core medicines, and user testing for bio-based materials.Yim added, "The ministry is ensuring that preferential drug pricing for the use of domestic raw materials and essential medicines is reflected in the drug price system reform plan. The goal is to create a structure that links budget support not only to simple raw material procurement but also to the improvement of facilities and equipment. Enhancing overall supply chain stability remains the core of this year's projects."Yim emphasized, "The issue of localizing and increasing the self-sufficiency rate of raw materials and bio-based components has been repeatedly pointed out during parliamentary audits," and added, "The MOHW will establish a research commission to develop a roadmap for API self-sufficiency and identify additional tasks through a two-track strategy that links the budget to pharmaceutical policies."
- Company
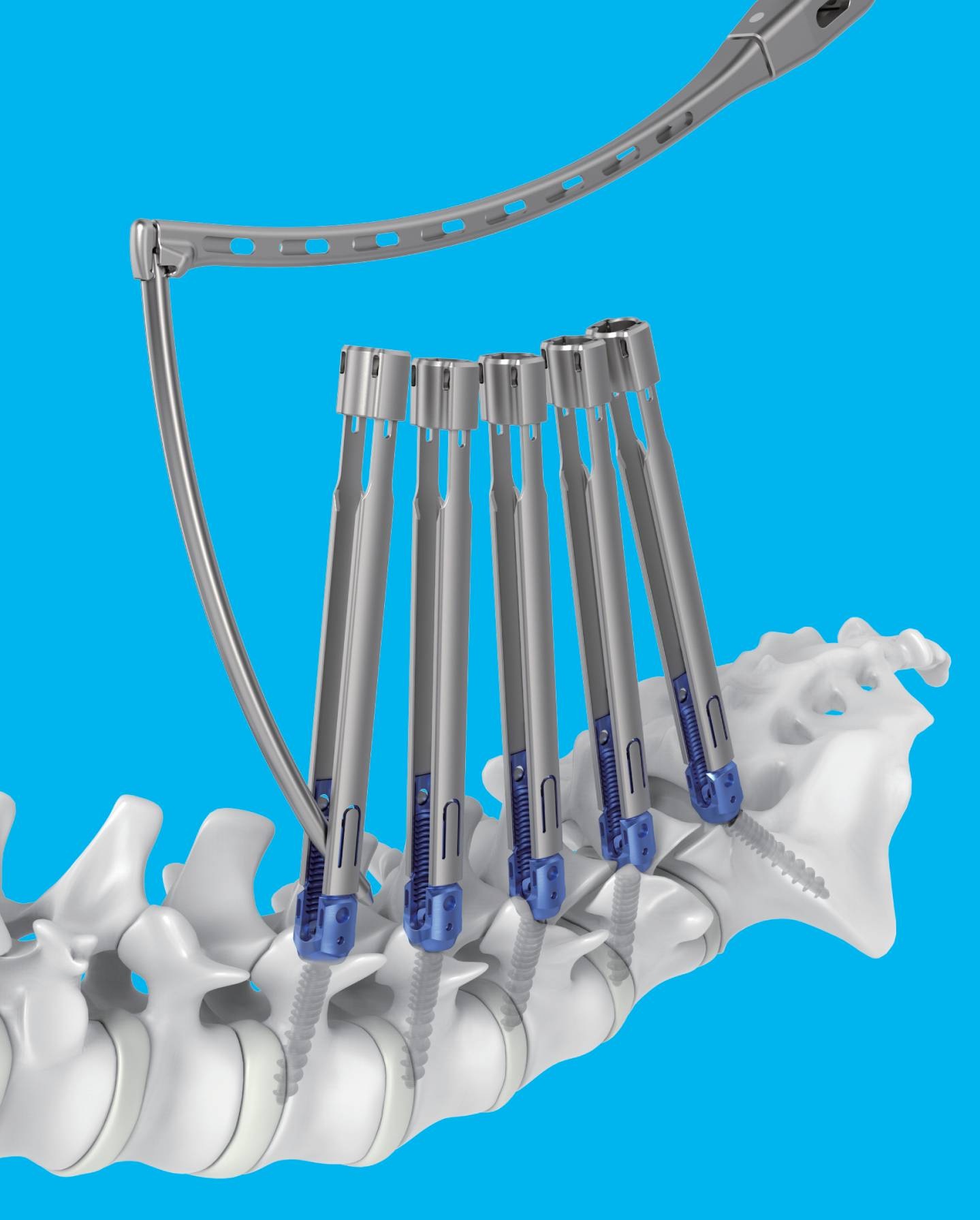
- Medtronic Korea launches new spine portfolio, Kanghui
- by Hwang, byoung woo Jan 22, 2026 08:22am
- 메드트로닉 신규 척추 포트폴리오 강휘Medtronic Korea (CEO Seung-rok Yoo) announced on the 21st that it has launched a new spine portfolio, “Kanghui,” aimed at expanding access to spine solutions.Kanghui is a new brand designed to deliver Medtronic’s accumulated technological expertise and quality management capabilities built across a broad range from the cervical spine to the lumbar spine at more affordable price points.With the introduction of Kanghui, Medtronic has established a comprehensive spine portfolio in Korea, ranging from standard models to premium models.The first product introduced within the Kanghui portfolio is the ‘ECO MIS’ system, an interbody fixation device.It consists of screws and rods in various shapes and lengths, along with fixation set screws, to provide patients with the appropriate size, shape, and design.It is used for minimally invasive surgery to stabilize and fixate the posterior thoracic, lumbar, or sacral spine in patients with unstable spinal fractures, spinal deformities (such as scoliosis), degenerative spinal disorders, spinal stenosis, degenerative disc disease (such as disc herniation), and segmental instability.Seung-rok Yoo, CEO of Medtronic Korea, said, “The introduction of Kanghui is part of a customized portfolio strategy reflecting the growing number of spinal disease patients in Korea and the expansion of specialized spine hospitals. Medtronic will continue to optimize and expand its portfolio to strengthen its competitiveness in the domestic spine market, while also contributing to the advancement of surgical techniques and the creation of a better treatment environment through partnerships with Korean medical professionals.”Meanwhile, the number of spinal disease patients in Korea increased by 9.1% over five years, from 8,912,158 in 2020 to 9,723,544 in 2024. The number of spinal surgery patients increased by 16.7% during the same period.Medtronic is a market leader with a total spine solution portfolio covering spinal disorders across the entire spine, from the cervical to the lumbar region.Through its extensive portfolio, which includes spinal fixation devices (Solera, Infinity, Zevo), minimally invasive spinal fixation systems (Sextant II, Longitude II), interbody fusion devices (Capstone, Clydesdale), and artificial discs (Prestige LP), it provides diverse minimally invasive treatment options.In addition, Medtronic integrates advanced technologies such as surgical navigation systems, contributing to improved surgical precision and enhanced patient safety.
- Company
- Drug price cuts without support disturb pharma independence
- by Hwang, byoung woo Jan 22, 2026 08:22am
- The Korean pharmaceutical industry has voiced strong concerns regarding the government's proposed drug price reform plan.Dailypharm 55th New Year Special Future ForumIt is a warning that repeated drug price cuts are depleting the foundational R&D strength of the domestic pharmaceutical industry and could ultimately lead to a health security crisis, such as the discontinuation of essential medicine supplies.On the 21st, DailyPharm hosted its 55th New Year Special Future Forum at the main auditorium of the The Catholic University of Korea Institute of Biomedical Industry under the title, "Our Attitude in Facing the Era of Great Transformation in Drug Pricing," to collect frontline opinions.The forum was attended by key figures including Yeon-sook Kim, Head of the Division of Pharmaceutical Benefits at the Ministry of Health and Welfare (MOHW); Jun-seop Park, Board of Director at Jeil Pharmaceutical; Jaeho Jung, Head of Department at Novartis Korea; and members of the Healthcare Team from the law firm Lee & Ko. They shared various views on the contents, analysis, and response strategies related to the drug price reform plan."KRW 63T in Cumulative Price Cuts… Unpredictability is the Greatest Risk"Jun-seop Park, Board of Director at Jeil Pharmaceutical, who delivered a presentation representing the domestic pharmaceutical industry, emphasized that repeated drug price reduction policies are forcing the industry to face a crisis of an unpredictable investment environment.According to Park, starting with the introduction of the actual transaction price reimbursement system in 1999, followed by the 2012 blanket price cuts and the 2020 differential requirement system, continuous reductions through 2023 are estimated to have resulted in a cumulative decrease of approximately KRW 63 trillion.The industry's greatest concern regarding these continuous cuts is 'uncertainty.'Park said, "This unpredictable policy environment makes it virtually impossible for pharmaceutical companies to establish long-term survival strategies."However, it was also emphasized that despite the repeated price-cutting environment, the domestic pharmaceutical industry has continued to build achievements in new and incrementally modified drugs (IMDs).Jun-seop Park, Board of Director at Jeil PharmaceuticalAccording to growth indicators for 2024 compared to 2000 presented by Director Park: ▲ Industry scale: KRW 7.9 trillion → KRW 29.8 trillion (277% growth); ▲ Number of employees: 55,000 → 120,000 (118% growth).In particular, R&D investment surged 18-fold (1727%) from KRW 0.197 trillion to KRW 3.6 trillion. Since the launch of the first domestically developed new drug in 1999, the industry has produced 41 new drugs and 142 IMDs, establishing advanced clinical and quality systems.However, Park warned that this virtuous cycle of growth is coming to a halt.As the basis, he cited the "essential medicine supply discontinuation" crisis. According to data from the Ministry of Food and Drug Safety (MFDS), the number of reported cases of drug supply discontinuation and shortages has nearly tripled over the five years since 2020."While prices have risen by 20%, the standards for low-priced medicines have been frozen for over 10 years, threatening the sustainability of manufacturing basic medicines," Park said. "In Europe, unsustainable pricing policies are also acting as one of the primary causes of drug shortages."The issue of equity in government support was also brought to the fore. While the government's R&D investment ratio for all industries stands at 21–24%, the ratio for the pharmaceutical industry was a mere 5.5% as of 2023."While companies cover 94.2% of pharmaceutical R&D with their own profits, slashing the profitability of generics and IMDs, which form that financial base, is like cutting off a runner's supply line during a marathon for new drug development," Park compared."Instead of short-term indicators like the R&D investment ratio of the past three years, we must comprehensively evaluate cumulative investment amounts, continuity, and substantial outcomes," Park suggested. "It is time to consider drug price preferences through the designation of 'Research-Oriented Pharmaceutical Companies' for those who continue substantial investment, in addition to the existing 'Innovative Pharmaceutical Companies' certification."Park urged, "What the pharmaceutical industry needs right now is not drug price cuts, but the breathing room and support to prepare for the next 10 years. Instead of securing finances by cutting drug prices, we need policies that strengthen self-sustainability, allowing the industry to create jobs and reinvest in global competitiveness through appropriate drug pricing.""Cutting Generic Prices to Approximately 40%, Legal Rationality in Question"Following this, Attorney Jin-hwan Jeong from the law firm Lee & Ko presented a legal and industrial analysis of the drug price reform plan announced by the MOHW last November.Member of the Healthcare Team from the law firm Lee & KoHe also questioned the government's plan for drug price cuts related to the generic drug price estimation criteria."The government cites cases from Japan and France, but it is worth considering whether a horizontal comparison is possible given the industrial structures and the proportion of global new drugs in those countries," Jeong said. "We need to examine whether there is rationality in how a cut to the 40% range contributes to stimulating new drug development by domestic pharmaceutical firms."Furthermore, he raised questions regarding the 'differentiation of the price addition system,' a core part of the reform. The government's plan grants high price additions only to the top 30% of Innovative Pharmaceutical Companies in terms of R&D ratios."It is difficult to see a substantial difference in R&D capabilities between a company at the 30th percentile and one at the 31st, and a structure where rankings change every year based on sales fluctuations hinders corporate predictability," Jeong said. "Evaluating companies primarily by ratios without considering comprehensive indicators, such as cumulative R&D investment or technology transfer achievements, may be irrational."Jeong added, "There may be grounds for claims regarding the deviation or abuse of discretionary power under the Administrative Litigation Act. The collapse of an industry happens in an instant, but its revival takes a long time. We need a detailed institutional design where welfare and industry can coexist."Predictability over Speed… Issues of Scope and Timing for Listed DrugsIn the subsequent Q&A session, questions focused on the 'speed' and 'predictability' of the policy.Attendees expressed concern that, as the reform is being pushed forward rapidly, a 'big picture timeline' for when and to what extent the system will be applied must be presented first so companies can prepare.There were also questions reflecting anxiety about the uncertainty of policy implementation, specifically about which of the currently listed medicines will be subject to price cuts and when.(from left) Yeon-sook Kim, Head of the Division of Pharmaceutical Benefits at the Ministry of Health and Welfare (MOHW), ; Jaeho Jung, Head of Department at Novartis Korea, Jun-seop Park, Board of Director at Jeil PharmaceuticalThe standard view among the domestic pharmaceutical industry was that, since each company had already established mid- to long-term business plans, an accelerated implementation date could destabilize management, employment, and decisions on development and investment.In response, Yeon-sook Kim, Head of the Division of Pharmaceutical Benefits at the MOHW, expressed a commitment to setting specific targets as soon as possible to increase industry predictability."The government also fully recognizes that policy predictability is important," Director Kim explained. "We will quickly finalize and announce the targets for listed drug adjustments, and we are reviewing measures for a soft landing, considering the impact on the industry."Kim emphasized that rather than a unilateral speed race, the government would ensure detailed adjustments through communication with the industry, stating, "We will not focus simply on cutting prices, but will sufficiently examine areas for supplementation by taking the voices of the industrial field into account."In conclusion, Kim added, "We are considering a phased implementation, starting with a primary review of medicines listed before 2012. We plan to finalize the specific targets and scope through technical verification and close communication with associations to avoid confusion."
- Policy
- MFDS reviews mutagenicity of clarithromycin impurity
- by Lee, Tak-Sun Jan 21, 2026 09:07am
- While investigations into nitrosamine impurities in the antibiotic clarithromycin are underway, the Ministry of Food and Drug Safety (MFDS) is reportedly also reviewing their mutagenicity.The European Medicines Agency (EMA) has already concluded that the clarithromycin-related nitrosamine impurity is non-mutagenic.If the MFDS also concludes that there is no mutagenicity, the threshold for excessive nitrosamine detection in clarithromycin would be removed, making quality control significantly easier for the pharmaceutical industry.According to the MFDS on the 20th, nitrosamine impurities (N-nitroso-N-desmethyl clarithromycin) were detected in excess in finished drug products using clarithromycin ingredients from India. Consequently, the MFDS has ordered an investigation into 77 companies holding related items.The MFDS orders an investigation within one month for known impurities and a three-month investigation for relatively unknown impurities.The threshold for nitrosamine impurities in clarithromycin has already been established. In December 2023, the acceptable intake was set at 1,500 ng per day.An official from the MFDS stated, "Impurities were first detected in raw materials from India, and excessive amounts were also confirmed in one domestic finished drug product using those materials. We have therefore issued investigation orders to all companies with relevant items."Upon receiving reports of the investigation results from these companies, the MFDS will review usage patterns and supply status before ordering product recalls.However, it has been noted that the MFDS is also reviewing the mutagenicity (a chemical reaction to cause genetic mutations) of the clarithromycin nitrosamine impurity.While the European EMA has already concluded it is non-mutagenic, the U.S. FDA is currently conducting review.If the impurity is determined to be non-mutagenic, it would no longer pose a regulatory issue, even if detected in excess, since the acceptable limit would be deleted.The MFDS official explained. "We are conducting the mutagenicity review and the impurity investigation concurrently," adding, "If the conclusion that clarithromycin nitrosamine is non-mutagenic had reached first, this investigation would not have taken place."Meanwhile, clarithromycin is a widely used antibiotic for infections of the upper and lower respiratory tracts.